Serum levels of glucose and lactate in patients treated under total intravenous anesthesia with propofol-fentanyl and under balanced anesthesia with isoflurane-fentanyl
Zlata Mujagić
[*]
[1]
Elsada Čičko
[2]
Vesna Vegar-Brozović
[3]
Mirsada Prašo
[2]
Introduction
Stress response to surgery is modulated by several factors, including severity and duration of surgical trauma, surgical technique, and type of anesthesia. Data on the effects of anesthetic and analgesic regimens upon metabolic responses to surgery in humans are varying and even controversial. The biochemical factors initiating, regulating and sustaining the metabolic response to surgery have not been fully identified.
Changes in the protein and glucose metabolism during and after surgery have been documented (1). Endogenous amino acid oxidation and amino acid release from the muscle after abdominal surgery have been shown to increase (1). Hyperglycemia is a prominent feature of the metabolic response induced by surgical trauma (2). Hyperglycemia is related to the degree of surgical trauma and can be, probably, influenced by the anesthetic technique. Whereas inhaled anesthetics exert only minimal inhibitory influence on the surgical stress response, propofol anesthesia attenuates the intraoperative increase in plasma glucose (3,4).
The effects of surgical tissue trauma and type of anesthesia on the immediate metabolic response have not yet been fully clarified. Hence, the aim of the present study was to investigate the effect of total intravenous anesthesia (TIVA) with propofol-fentanyl and the effect of balanced anesthesia with isoflurane-fentanyl on the circulating levels of glucose and lactate in patients subjected to elective low abdominal surgery.
Materials and methods
Patients
The prospective study included 50 patients (22 male and 28 female, Caucasians, residents of the narrow region in Tuzlasurrounding). All of them were subjected to the low abdominal surgery (40 colon cancer operations and 10 hysterectomies) at University Department of Surgery and Traumatology and University Department of Gynecology, Tuzla University Clinical Center, Tuzla, Bosnia and Herzegovina.
The inclusion criteria for patients were as follows: elective low abdominal surgery; age between 35 and 60 years; ASAI/II status (American Society of Anesthesiologists I/II classification); almost the same length of the surgery, and almost the same degree of surgical trauma.
Exclusion criteria were as follows: presence of a metabolic, hepatic or renal disease, and receiving any medication known to affect glucose metabolism.
The study was approved by the Council of the School of Medicine, University of Tuzla. All patients signed informed consent forms before inclusion in the study.
Patients were randomly divided into two groups: experimental group of 25 ASA I/II patients treated under TIVA with propofol-fentanyl, and control group of 25 ASA I/II patients treated under general balanced anesthesia with isoflurane-fentanyl.
Anesthesia protocol
The protocol of balanced anesthesia was as follows: premedication with midazolam 0.1 mg/kg i.m.; induction with thiopental sodium 5 mg/kg, with fentanyl 0.1 mg before intubation with the overall dose of 0.005 mg/kg before surgical incision; maintenance of anesthesia with isoflurane 1.2-2.4 vol% mixed with nitrous oxide and oxygen at a ratio of 1:1; suxamethonium hydrochloride 1.5 mg/kg and Tracrium 0.5-0.8 mg/kg for intubation and myorelaxation.
The protocol for TIVA was as follows: premedication with midazolam 0.1 mg/kg i.m.; induction with propofol 2 mg/kg and fentanyl 0.1 mg before intubation with the overall dose of 0.005 mg/kg before surgical incision; maintenance of anesthesia with propofol infusion 6-12 mg/kg/h with ventilation by intermittent positive pressure (IPPV) with a mixture of air, oxygen and FiO2 33-50%; repetitive fentanyl doses of 0.1 mg depending on clinical parameters; suxamethonium hydrochloride 1.5 mg/kg and Tracrium 0.5-0.8 mg/kg for intubation and myorelaxation.
Hemodynamic monitoring (heart rate, systolic and diastolic blood pressure) and expired air CO2 monitoring were performed in all patients during surgical treatment. Patients received electrolyte-balanced solution during the operation, and Ringer-lactate solution and 5% glucose solution 24 hours after the operation. Heart rate, blood pressure and pupillary dilatation were relevant parameters for the anesthesia course follow up. Anesthetics and myorelaxants were added according to the protocol during the operation.
Samples
Blood samples for glucose and lactate measurements were drawn at exact time points: 30 minutes before the beginning of the surgery (T0), 30 minutes after the beginning of the surgery (T1), at the end of the surgery (T2), 2 hours after the surgery (T3), and 24 hours after the surgery (T4). Serum levels of glucose were measured using a commercially available Glucose FlexTM kit (Dade Behring). Serum levels of lactate were measured using a commercially available Lactic Acid FlexTM kit (Dade Behring).
Statistical analysis
The distribution of quantitative variables was determined by using distribution histograms with probability plots and de-trended probability plots. The results were evaluated with nonparametric Mann-Whitney test, with mean values, standard deviations (SD), and standard errors (SE) calculated. The value of P = 0.05 was considered statistically significant. All statistical analyses were performed with SPSS statistical software, version 10.
Results
According to the kits used for glucose and lactate determination, the normal range for glucose was 3.9-6.1 mmol/L, and for lactate 0.4-2.0 mmol/L. The mean serum concentrations of glucose measured at time points T1,T2, and T3 were all above the normal range for glucose in patients treated under balanced anesthesia with isoflurane-fentanyl, while the T0 and T4 values were within the normal range. Only the T2 glucose concentration was above the normal range in patients treated under TIVA with propofol, while other values were within the normal range for glucose.
The mean serum level of lactate measured at T2 was above the normal range in patients treated under balanced anesthesia, while lactate values at other points were within the reference range. Lactate concentrations measured at T1, T2, T3 and T4 were within the normal range in patients treated under TIVA with propofol.
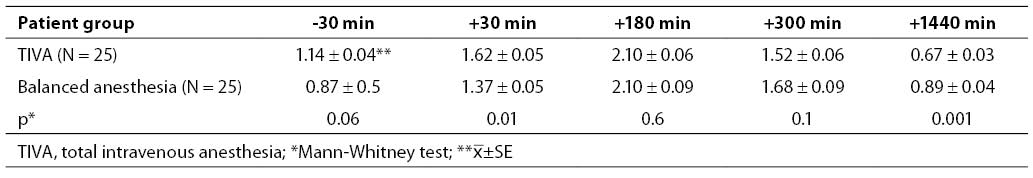
The mean serum levels of glucose measured at T1,T2, and T3 time points in patients operated under TIVA with propofol-fentanyl were significantly lower than those in patients treated under general balanced anesthesia with isoflurane-fentanyl (P = 0.03, P = 0.001 and P < 0.001, respectively) (Table 1).
The mean circulating levels of lactate measured at T4 in TIVA group of patients was significantly lower in comparison with that in patients treated under balanced anesthesia with isoflurane (P = 0.001), while serum lactate determined at T1 point in the balanced anesthesia group was lower than that in TIVA group (P = 0.01) (Table 2).
Table 1. Circulating glucose levels (mmol/L) in patients treated under TIVA with propofol-fentanyl and patients treated under balanced anesthesia with isoflurane-fentanyl at different time points
Table 2. Circulating lactate levels (mmol/L) in patients treated under TIVA with propofol-fentanyl and patients treated under balanced anesthesia with isoflurane-fentanyl at different time points
Discussion
The results of this study showed the mean serum concentrations of glucose and lactate during the operation and shortly after the operation to be significantly lower in patients treated under TIVA with propofol-fentanyl in comparison with those in patients treated under general balanced anesthesia with isoflurane-fentanyl (Tables 1 and 2). The results suggested the metabolic response to surgery to be probably attenuated and thus improved in patients treated under TIVA with propofol-fentanyl as compared with that in patients treated under general balanced anesthesia with isoflurane-fentanyl. Results of some other similar studies are comparable with those obtained in our study. Propofol anesthesia supplemented with sufentanil, in contrast to inhaled anesthesia, suppressed the intraoperative increase in plasma glucose concentration (4). Inhibitory effects of propofol on the sympathoadrenal system have been documented in patients undergoing cardiac surgery (5), and documented in vitro when propofol concentrations, similar to those observed during the induction of anesthesia, decreased the basal and nicotine-stimulated release of catecholamines from chromaffin cells (5). TIVA with propofol blunts perioperative metabolic and endocrine response when compared to inhaled anesthesia with sevoflurane (6). Intraoperative plasma concentrations of glucose, lactate and free fatty acids were significantly lower in TIVA in comparison with inhalation anesthesia (7). Anesthetic agents can affect glucose metabolism through, at least partly, sympathetic tone modulation.
Intraoperative glucose levels and that measured two hours after the surgery in our isoflurane-fentanyl anesthesia group were above the normal range for glucose; however, in patients treated under TIVA with propofol, only T2 glucose concentration was above the normal range while the levels measured at other points were within the normal range. Lactate levels measured at all points in both patient groups, except for T2 in balanced anesthesia, were within the reference range. These findings are in accordance with some other literature reports (4,8-12).
It has been proposed that the mechanisms by which the metabolic effects of anesthesia are mediated are likely to be hormonal (8). The stress hormone cortisol is a powerful promoter of gluconeogenesis in the liver (13), and acts on the glycogen storage in the liver as well as on the reduction of glucose utilization in peripheral tissues. Growth hormone and prolactin have a role in stress, probably by their hyperglycemic action in the liver (13,14). Propofol anesthesia did not significantly affect whole body protein synthesis and oxidation but caused a small, although significant, decrease in the whole body protein breakdown, possibly mediated through the suppression of plasma cortisol concentration (15). Furthermore, some data indicate that anesthetic concentrations of propofol inhibit O2- generation by D-glucose overload, and that this may occur via mechanisms that include, at least in part, the inhibition of cellular glucose uptake (16).
Protein degradation in skeletal muscle, glycolysis, and gluconeogenesis are the prominent characteristics of intermediary metabolism in patients under surgical stress conditions. Direct relationship between the whole body protein breakdown and glucose production in surgical patients have been observed (3,9,17). Muscle proteins are broken down to provide gluconeogenic amino acids for de novo gluconeogenesis in the liver. Promotion of gluconeogenesis by cortisol in the liver is caused primarily by the stimulation of protein catabolism (13,18). Kinetics studies of protein and glucose metabolism during abdominal surgery have revealed depression of the whole body protein and glucose metabolism with the hyperglycemic response being caused by a decreased whole body glucose clearance and a significant relationship between glucose production and protein breakdown (9). The rate of the overall metabolism is increased in stress conditions, but the capacity of oxidative metabolism is limited. This is one of the main reasons why catabolic pathways begin to work unaerobically and to produce, among other metabolites, lactate.
Results obtained in our study showed that serum levels of glucose and lactate were increased during surgical treatment, and that this increase was more pronounced in patients treated under general balanced anesthesia with isoflurane than in those under TIVA with propofol. The observed changes are probably due to surgical stress, protein catabolism and gluconeogenesis. However, only two metabolic parameters were investigated in the two anesthesia groups of our patients. It was a major limitation to our study because it was not possible to precisely evaluate which process was more responsible for the control of glycemia in TIVA group of patients.
Metabolic response to surgery is probably attenuated and thus improved in patients treated under TIVA with propofol in comparison with that in patients treated under general balanced anesthesia with isoflurane. Our results are in accordance with similar data reported in the relevant literature, and they indicate that multimodal approaches combining anesthetic, analgesic and surgical strategies will result in better control of glycemia during the intra- and perioperative periods, and thus in improvement of the clinical outcome of disease.
Acknowledgments
We thank Prof. Hamza Mujagić, MD, PhD, for his very useful suggestions in the study design and statistical analysis of the results. We also thank the staff of the University Department of Surgery, University Department of Gynecology, and Department of Biochemistry, Tuzla University Clinical Center, Tuzla, Bosnia and Herzegovina, for their technical assistance during the study.
References
1. Schricker T. The catabolic response to surgery: how can it be modified by the anesthesiologist? Can J Anesth 2001;48:R1-R5.
2. Schricker T, Lattermann R, Schreiber M, Geisser W, Georgieff M, Radermacher P. The hyperglycaemic response to surgery: pathophysiology, clinical implications and modification by the anaesthetic technique. Clin Intern Care 1998;9:118-28.
3. Kocamanoglu IS, Sahinoglu AH, Tür A, Baris S, Karakaya D. The comparison of the effects of TIVA and inhalation anaesthesia on hemodynamic conditions, metabolic-endocrine response to trauma and muscle relaxant consumption. Turk Anestez Reanim 2000;28:452-6.
4. Schricker T, Carli F, Schreiber M, Wachter U, Geisser W, Lattermann R, et al. Propofol/sufentanil anesthesia suppresses the metabolic and endocrine response during, not after, lower abdominal surgery. Anesth Analg 2000;90:450-5.
5. Ng A, Tan SSW, Lee HS, Chew SL. Effect of propofol infusion on the endocrine to cardiac surgery. Anaesth Intens Care 1995;23:543-7.
6. Kas D, Gönüllü M, Kol IÖ. The effect of different anaesthetic techniques on stress response to the surgery. Turk Anestez Reanim 2005;33:471-9.
7. Crosier TA, Sumpf E. The effect of total intravenous anesthesia with S-(+)-ketamine/propofol on hemodynamic, endocrine and metabolic stress reactions in comparison to alfentanil/propofol in laparotomy. Anaesthesist 1996;45:1015-23.
8. Lattermann R, Schricker T, Wachter U, Georgieff M, Goertz A. Understanding the mechanisms by which isoflurane modifies the hyperglycaemic response to surgery. Anesth Analg 2001;93:121-7.
9. Schricker T, Lattermann R, Fiset P, Wykes L, Carli F. Integrated analysis of protein and glucose metabolism during surgery: effects of anesthesia. J Appl Physiol 2001;91:2523-30.
10. Schricker T, Galeone M, Wykes L, Carli F. Effect of desflurane/remifentanil anaesthesia on glucose metabolism during surgery: a comparison with desflurane/epidural anaesthesia. Acta Anaesthesiol Scand 2004;48:169-73.
11. Lattermann R, Wachter U, Georqieff M, Goertz A, Schricker T. Catabolic stress response during and after abdominal surgery. Comparison between two anesthesia procedures. Anaesthesist 2003;52:500-6.
12. Geisser W, Schreiber M, Hofbauer H, Lattermann R, Fussel S, Wachter U, et al. Sevoflurane versus isoflurane – anaesthesia for lower abdominal surgery. Effects on perioperative glucose metabolism. Acta Anaesthesiol Scand 2003;47:174-9.
13. Mathews CK. Biochemistry. San Francisco: Addison Wesley Longman; 1999.
14. Devlin TM. Textbook of biochemistry with clinical correlations. New York: Wiley-Lyss; 1997.
15. Schricker T, Klubien K, Carli F. The independent effect of propofol anesthesia on whole body protein metabolism in humans. Anesthesiology 1999;90:1636-42.
16. Karashima Y, Oike M, Takahashi S, Ito Y. Propofol prevents endothelial dysfunction induced by glucose overload. Br J Pharmacol 2002;137:683-91.
17. Schricker T, Wykes L, Carli F. Epidural blockade improves substrate utilisation after surgery. Am J Physiol Endocrinol Metab 2000;279:E646-E53.
18. Burtis CA, Ashwood ER, eds. Tietz textbook of clinical chemistry. Philadelphia: Saunders; 1999.