Introduction
Based on results of past investigations, the levels of serum lipids including triglycerides, total cholesterol, high density lipoprotein cholesterol (HDL-cholesterol) and low density lipoprotein cholesterol (LDL-cholesterol), were in significant correlation with numerous psychopathological conditions like schizophrenia, depression, PTSD, and other anxiety disorders, aggressiveness, impulsiveness and suicide (1-6).
Cholesterol has several important functions in the central nervous system. It is a constituent of the neuronal membrane, and plays a significant role in the process of neurotransmission and in the second messenger system in the brain (7). Low cholesterol level is believed to enhance the risk of depression due to the neuronal dysfunction occurring because of changes in microviscosity of the cellular membrane or disorders in signal transduction (8-9). Most past studies have indicated a correlation between depression symptoms and serum cholesterol level. Low cholesterol concentration was observed in depressive patients in some studies (7,9-13), while in others this finding was not confirmed (14). Steegmans et al. observed significantly elevated risk of depression symptoms in middle-aged men with chronically lowered serum cholesterol concentration (15). As depression is one of the major causes of suicide, the studies of association between low cholesterol and depression may be of substantial importance, which was indicated by a study describing a correlation between cholesterol level and suicidality (8).
Although the association between serum total cholesterol level and depression symptoms has been frequently investigated, few studies described the correlation of other lipids and depressive disorder (16). Thus, one study established a significantly lower concentrations of serum cholesterol, HDL-cholesterol, and cholesterol/HDL-cholesterol ratio in patients with depressive disorder in comparison to a healthy control group (17). Results of another study demonstrated considerable differences in the levels of triglycerides, very low density lipoprotein cholesterol (VLDL-cholesterol), and HDL-cholesterol between melancholic and atypical depression (16). Also, one study found a significantly lower LDL-cholesterol level in depressive patients than in the control group, but no such difference was recorded for the level of triglycerides (12).
Despite the fact that a large number of studies of lipids in depressive disorder have been conducted, there have been only several reports that considered differences in serum lipid concentrations according to individual depression subtypes, and these yielded contrary results. Thus, in some studies no differences were found in serum lipid levels of patients with melancholic and non-melancholic depression (17,18), or in patients with melancholy and atypical depression (19), while Huang TL et al. in their study established a significant difference in the concentrations of triglycerides, VLDL-cholesterol, and HDL-cholesterol in patients affected by melancholy and atypical depression (16).
The aim of this study was to examine the concentrations of serum lipids (cholesterol, triglycerides, HDL-cholesterol, LDL-cholesterol, VLDL-cholesterol) and lipid fraction ratios (cholesterol/HDL-cholesterol and LDL-cholesterol/HDL-cholesterol) in relation to a type of depressive disorder (melancholic, atypical and dysthymic).
Materials and methods
Subjects
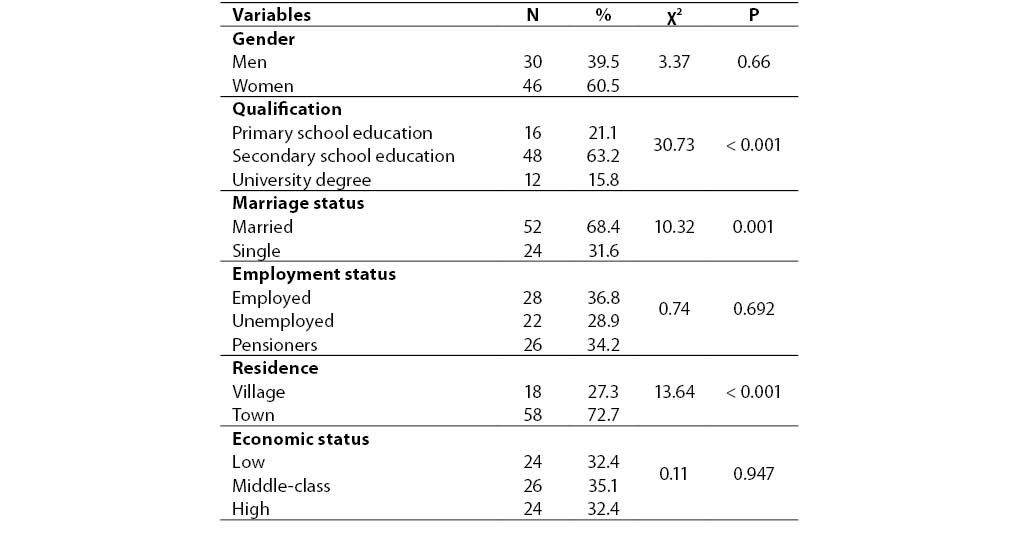
The study included 76 subjects affected by depressive disorder and hospitalized in Sestre milosrdniceUniversity Hospital during a 12-month period in years 2005 and 2006. The patient sample consisted of 30 men and 46 women, mean age ± SD = 53 ± 10 years, mean body mass index (BMI) (mean ± SD) 25.66 ± 4.29 kg/m2. Mean age ± SD at disease onset was 45 ± 10 years, while disease duration (mean ± SD) was 8 ± 6 years with mean number of hospitalizations (mean ± SD) 3.11 ± 2.55. Patient sociodemographic characteristics are shown in Table 1.
Table 1. Sociodemographic characteristics of patients with depressive disorder who participated in the study.
Subjects were treated pharmacologically by antidepressants (fluoxetine, fluvoxamine, paroxetine, sertraline, clomipramine) and anxiolytics (alprazolam, oxazepam, diazepam, clonazepam). None of the psychopharmaceutics above has any effect on serum lipid concentrations (20). None of the subjects had been treated by hypolipemics prior to their participation in the study. Criteria for exclusion from the study were other concurrent psychiatric disorders, drug addiction, alcoholism, hypertension, cardiovascular diseases, thyroid disorders, diabetes mellitus, lipoprotein metabolism disorders, nutrition disorders and organic brain syndromes. Since dietary habits and physical activity may affect test results, all tests were conducted between days 14 and 16 of hospital stay, i.e. during the period of uniform diet and level of physical activity of all subjects.
All subjects signed their written consent for participation in the study, and the study was approved by a competent ethical committee.
Diagnostic instruments and formation of subject groups
Diagnosis of depressive disorders was made according to criteria for depressive disorders of the Diagnostic and Statistical Manual for mental disorders, 4th revision (DSM IV) (20), and application of the Hamilton Rating Scale for Depression (HAMD-17) (22). Depression subtype (melancholic, atypical, dysthymia) was diagnosed using MINI questionnaire based on DSM IV criteria (21, 20). Subjects were divided into groups according to depression type into three different diagnostic categories: a group with melancholic characteristics, a group with atypical characteristics, and a group with dysthymia.
Laboratory tests and anthropometric measurements
Blood samples were taken from the cubital vein into glass vacuum test-tubeswithout an anticoagulant at 8-9h a.m. after minimum 12h overnight fast and 30 min rest immediately prior to sample collection. Serum concentrations of cholesterol, triglycerides and HDL-cholesterol were determined by enzymatic method immediately after sample collection using commercial laboratory kits (Olympus Diagnostic, GmbH, Hamburg, Germany) on Olympus AU 600 automated analyzer. Interassay coefficients of variation in the laboratory were 3.2% for cholesterol, 2.5% for triglycerides, and 3.0% for HDL-cholesterol. LDL-cholesterol and VLDL-cholesterol levels were calculated by conventional formulas from triglyceride, cholesterol, and LDL-cholesterol levels (LDL-cholesterol = cholesterol - triglycerides /2.2 - HDL-cholesterol; VLDL-cholesterol = triglycerides/5). Recommended laboratory intervals for the measured parameters were: cholesterol < 5.0 mmol/L, LDL < 3.0 mmol/L, HDL > 1.0 mmol/L, VLDL-cholesterol < 1 mmol/L, LDL/HDL ratio < 4.0, cholesterol/HDL-cholesterol ratio < 2.5, and triglycerides < 1.7 mmol/L. The height and weight of each patient, who were barefoot and in light clothes, were measured in standing position on a medicalscale that measures height and weight. Body mass index (BMI) was calculated by dividing kilograms by squared height in meters.
Statistical data processing
Data were stored in MS Access 2000 database, and SPSS statistical program (SPSS for Windows 11.0, SPSS, Chicago, IL, USA) was used for statistical analysis. Descriptive statistical methods were used in data processing. Hi-square test was used to assess differences in sociodemographic data. To test differences in serum lipid level between groups, one way analysis of variance (ANOVA) was used. Scheffeposthoc test was applied to examine differences between individual groups, while covariance analysis (ANCOVA) was used to control the effect of age and body mass index on investigated parameters. Sample distribution was tested by Kolmogorov-Smirnov test. P < 0.05 probability level was considered statistically significant.
Results
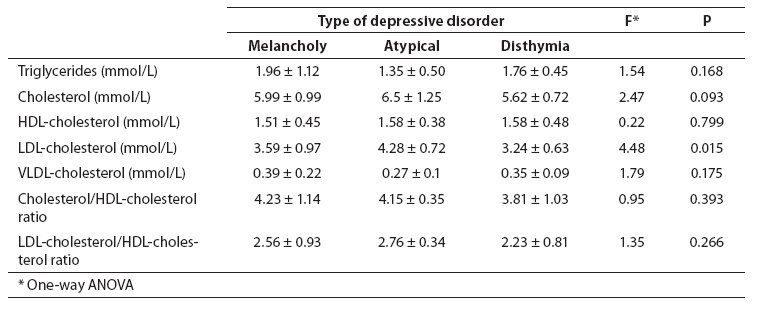
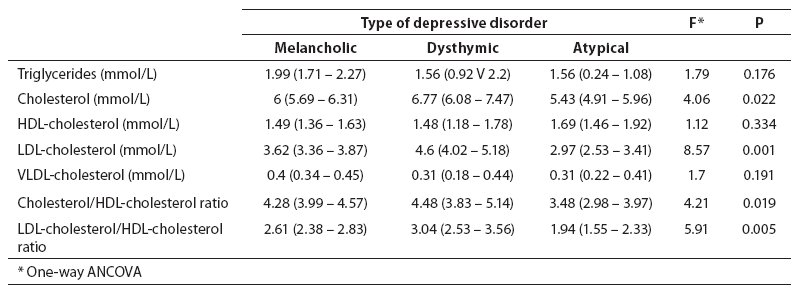
Table 2 showsserum lipid concentrations in relation to the type of depression. LDL-cholesterol level was significantly lower in patients with atypical depression than in dysthymic patients (F(2.63) = 4.48, P = 0.015, ANOVA; P = 0.016, Scheffe test), and this difference persisted after adjustments for age and BMI (F(2.61) = 8.57; P = 0.001, ANCOVA). One-way analysis of variance revealed no statistically significant difference in the levels of triglycerides, cholesterol, and cholesterol/HDL ratio and LDL-cholesterol/HDL-cholesterol ratio among patients with melancholic and atypical depression and dysthymia. However, after age and BMI adjustments, a statistically significantly lower total cholesterol concentration was found in patients with atypical depression compared to patients with melancholy and dysthymia (F(2.61) = 4.06; P = 0.022, ANCOVA), a significantly lower cholesterol/HDL-cholesterol ratio in patients with atypical depression than in melancholic and dysthymic patients (F(2.61) = 4.21; P = 0.019, ANCOVA), and finally a significantly lower LDL-cholesterol/HDL-cholesterol ratio in patients with atypical than in patients with melancholic depression and dysthymia (F(2.61) = 5.91; P = 0.005, ANCOVA) (Table 3).
Table 2. Serum lipids (mean ± SD) in patients with depressive disorder according to depression subtype (melancholic, atypical, disthymia).
Table 3. Serum lipids (mean (95% CI)) after age and BMI adjustment in patients with depressive disorder according to depression type (melancholic, atypical and dysthymic).
Discussion
Results of this study demonstrated significantly lower values of LDL-cholesterol, cholesterol, cholesterol/HDL-cholesterol ratio, and LDL-cholesterol/HDL-cholesterol ratio in patients with atypical depression compared to patients with melancholy and dysthymia. Also, the results of this investigation indicated no significant difference in the serum levels of triglycerides, HDL-cholesterol and VLDL-cholesterol among patients with atypical depression, melancholy, and dysthymia. A similar study conducted by Huang and Chen in 2004, where covariance analysis was used after age adjustment, suggested that serum concentrations of triglycerides, VLDL-cholesterol and HDL-cholesterol might be used as biological markers to distinguish depressive patients with atypical depression from patients with melancholic characteristics (16). Upon application of the same statistical analysis after adjustment for BMI in a similar study, no difference in serum lipid concentrations was found among subject groups with melancholic and atypical characteristics of depressive disorder (19). After application of the analysis of covariance and age and BMI adjustments, results of our study confirmed the levels of cholesterol, LDL-cholesterol, cholesterol/HDL-cholesterol ratio, and LDL-cholesterol/HDL-cholesterol ratio as possible biological markers for differentiation between certain clinical subtypes of depressive disorder.
Results of some studies demonstrated cholesterol/HDL-cholesterol ratio and LDL-cholesterol/HDL-cholesterol ratio (indication of atherosclerosis) to be of a significantly higher predictive value for the onset of coronary heart disease than cholesterol, HDL-cholesterol, and LDL-cholesterol concentrations (23). One of the relevant results of this study is the statistically significant difference in the above indices in patients with atypical, melancholic and dysthymic characteristics of depressive disorder, which indicates the possible existence of various degrees of risk for atherosclerosis progressionand consequent coronary heart diseasein patients with different clinical subtypes of depressive disorder.
Both elevated and decreased cholesterol levels may be related to serotonergic dysfunction. Primary decline in cholesterol level may lead directly to reduction in cerebral 5-HT activity by various mechanisms like changes in concentrations of 5-hydroxy triptamine (5-HT), of 5-HT receptor, and in 5-HT transporter activity. Moreover, low cholesterol level in depressive and suicidal individuals may be a consequence of disturbed free cholesterol esterification. A rise in cholesterol level may cause a decrease in 5-HT receptor sensitivity or in 5-HT transporter activity in depressive patients by direct binding to membrane receptors or transporter molecules, or indirectly by altering fluidity of the neuronal membrane. Based on past information, the cholesterol level could exert a considerable effect on the appearance of the clinical picture of depressive disorder, and on the response to psychofarmacological treatment. It seems that patients with low cholesterol level (< 4.0 mmol/L) are at an increased risk of suicide, while patients with elevated cholesterol level (> 5.6 mmol/L) more often exhibit resistance to treatment and more frequently have comorbid anxiety disorders (24).
In conclusion, the results of our study suggest that serum cholesterol- and LDL-cholesterol concentrations and cholesterol/HDL-cholesterol- and LDL-cholesterol/HDL-cholesterol ratios could serve as biological markers to distinguish between clinical subtypes of depressive disorder, implicating diagnostics, specifical psychofarmacological approach, and improved prevention of suicide. Drawbacks of this study are a small patient sample and the lack of a control group of healthy subjects. Also, we did not consider differences in dietary and smoking habits and alcohol consumption of our patients. It will be necessary to include larger subject samples in the healthy control group in further studies. Future studies will also require investigation of predictive values of cholesterol/HDL-cholesterol- and LDL-cholesterol/HDL-cholesterol ratios for the development of coronary heart disease in depressive patients according to different clinical subtypes.
Notes
Potential conflict of interest
None declared.
References
1. Chen CC, Lub FH, Wub JS, Chang CJ. Correlation between serum lipid concentrations and psychological distress. Psychiatry Res 2001;102:153-62.
2. Karlović D, Buljan D, Martinac M, Marcinko D. Serum lipid concentrations in Croatian veterans with post-traumatic stress disorder, post-traumatic stress disorder comorbid with major depressive disorder, or major depressive disorder. J Korean Med Sci 2004;19:765-71.
3. Marčinko D, Martinac M, Karlović D, Lončar Č. Cholesterol serum levels in violent and non-violent young male schizophrenic suicide attempters. Psychiatr Danub 2004;16:161–4.
4. Marčinko D, Martinac M, Karlović D, Filipčić I, Lončar Č, Pivac N, Jakovljević, M. Are there differences in serum cholesterol and cortisol concentrations between violent and non-violent schizophrenic male suicide attempters? Coll Antropol 2005;29:153–7.
5. Karlović D, Potkonjak J, Buljan D, Martinac M, Marčinko D. Serum lipids in patients with post-traumatic stress disorder, panic disorder, or generalized anxiety disorder. Acta Clinica Croatica 2004;43:9-14.
6. Karlović D, Martinac M, Buljan D, Zoričić Z. Relationship between serum lipid concentrations and posttraumatic stress disorder symptoms in soldiers with combat experiences. Acta Med Okayama 2004;58:23-7.
7. Ghaemi SN, Shields GS, Hegarty JD, Godwin FK. Cholesterol levels in mood disorders: high or low? Bipolar Disord 2000;2:60-4.
8. Golier JA, Marzuk PM, Leon AC, Weiner C, Tardiff K. Low serum cholesterol level and attempted suicide. Am J Psychiatry 1995;152:419-23.
9. Maes J, Delanghe J, Meltzer HY, Scharp´e S, D’Hondt P, Cosyns P. Lower degree of esterification of serum cholesterol in depression: relevance for depression and suicide research. Acta Psychiatr Scand 1994;90:252-8.
10. Morgan RE, Palinkas LA, Barret-Connor EL, Wingard DL. Plasma cholesterol and depressive symptoms in older men. Lancet 1993;341:75-9.
11. Glueck CJ, Tieger M, Kunkel R, Hamer R, Tracy T, Speirs J. Hypocholesterolemia and affective disorders. Am J Med Sci 1994;308:218-25.
12. Olusi SO, Fido AA. Serum lipid concentrations in patients with major depressive disorder. Biol Psychiatry 1996;40:1128-31.
13. Partonen T, Haukka J, Virtamo J, Taylor PR, Lonnqvist J. Association of low serum total cholesterol with major depression and suicide. Br J Psychiatry 1999;175:259-62.
14. Simons L, Simons J, McCallum J, Friedlander Y. Serum lipids and depression. Lancet 1993;341:1208.
15. Steegmans PHA, Hoes AW, Bak AAA, van der Does E, Grobbee DE. Higher prevalence of depressive symptoms in middle-aged men with low serum cholesterol levels. Psychosom Med 2000;62:205-11.
16. Huang TL, Chen JF. Lipid and lipoproteinlevels in depressive disorders with melancholic feature or atypical feature and dysthymia. Psychiatry Clin Neurosci2004;58:295-9.
17. Maes M, Smith R, Cristophe A, et al. Lower serum high density lipoprotein cholesterol (HDL-C) in major depression and in depressed men with serious suicidal attempts: relationship with immune-inflammatory markers. Acta Psychiatr Scand 1997;95:211-21.
18. Maes M, Smith R, Christophe A, Vandoolaeghe E, Van Gastel A, Neels H, et al. Lower serum high-density lipoprotein cholesterol (HDL-C) in major depression and in depressed men with serious suicidal attempts: relationship with immune-inflammatory markers. Acta Psychiatr Scand 1997;95:212-21.
19. Huang TL. Serum lipid profiles in major depression with clinical subtypes, suicide attempts and episodes. J Affect Disord 2005;86:75-9.
20. Stahl S. Essential psychopharmacology. Neuroscientific basis and practical applications. Cambridge: Cambridge University Press, 2002;199-295.
21. American Psychiatric Association. Diagnostic and Statistic Manual of Mental Disorders, IVth edition. Washington DC: American Psychiatric Association, 1994.
22. M.I.N.I. 5.0.0 (January 2000) Croatian version.
23. Hamilton M.A. Rating scale for depression. J Neurol Neurosurg Psychiatry 1960;23:56-62.
24. Sonnenberg LM, Quatromoni PA, Gagnon DR, Cupples LA, Franz MM, Ordovas JM, et al. Diet and plasma lipids in women. II Macronutrients and plasma triglycerides, high-density lipoprotein, and the role of total to high-density lipoprotein cholesterol in women: the Framingham nutrition studies. J Clin Epidemiol 1996;49:665-72.
25. Papakostas GI, Öngür D, Iosifescu DV, Mischoulon D, Fava M. Cholesterol in mood and anxiety disorders: review of the literature and new hypotheses. Eur Neuropsychopharmacol 2004;14:135-42.