Introduction
The first described cases of metabolic side-effects of antipsychotics date back to 1956. That was the time when, four years after introduction of the first antipsychoticchlorpromazine in therapy, data were published on the occurrence of hyperglycemia and glucosuria in previously euglycemic patients who were administered chlorpromazine. There were also concurrent descriptions of cases of impaired glycemic control in diabetics on chlorpromazine therapy. Upon discontinued administration of chlorpromazine, normalization of glycemia was achieved as well as diabetes control at the levels prior to antipsychotic therapy (1,2).
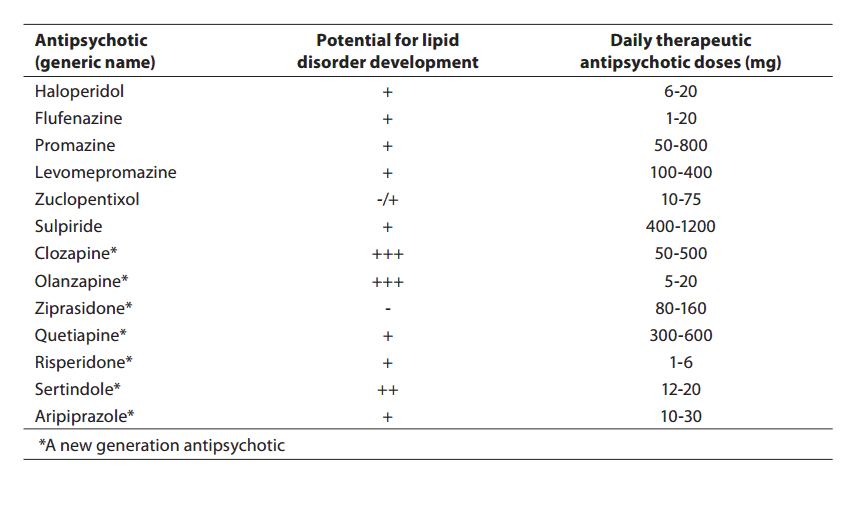
Metabolic side-effects have, however, been shown to accompany not only the administration of conventional antipsychotics like chlorpromazine. Actually, we presently face similar problems during introduction of the novel, so-called atypical antipsychotics. Introduction of atypical antipsychotics in therapy has significantly promoted the treatment of patients affected by schizophrenia and other psychotic disorders. Compared to conventional antipsychotics, the major advantage of these drugs is lower frequency of extrapyramidal side-effects and of hyperprolactinemia, and better overall tolerance. Still, some of atypical antipsychotics have been associated with body weight gain, occurrence of diabetes, and increase in cholesterol and triglyceride levels (Table 1).
Table 1. Review of the potential of some antipsychotics for the development of lipoprotein metabolism disorder
This review addresses differences in the effect of certain atypical antipsychotics on glucose and insulin homestasis and lipid metabolism, as well as the issue of the method of rational administration of antipsychotics accompanied by metabolic side-effects.
Effect of atypical antipsychotics on glucose and insulin homeostasis
The highest number of published clinical studies dealing with glucose and insulin homeostasis refer to clozapine, olanzapine and risperidone, with relatively few studies of metabolic side-effects of other atypical antipsychotics. When interpreting results of these studies, it should be taken into account that, in addition to administration of any of the above atypical antipsychotics to patients, there are at least other seven factors that may also contribute to the occurrence of tissue resistance to insulin and thus increase the risk for diabetes: old age, family history of type 2 diabetes, ethnic origin, obesity, smoking, lack of physical activity, and therapy with other diabetogenic drugs (3-5). Comparative studies have demonstrated that clozapine and olanzapine elevate the risk for occurrence of glucose intolerance and diabetes, while risperidone does not have such effect.
With regard to quetiapine, only two comparative studies have been published and they do not provide sufficiently clearresults. No comparative study has been published to include amilsulpride, zotepine, ziprasidone, and aripiprazole.
Clozapine
Melkersson et al. have measured insuline concentration after overnight fasting, and the concentration of clozapineand other classical antipsychotics. The study showed the insulin level to be positively correlated to clozapine concentration, while no correlation was observed between insulin level and the concentration of classical antipsychotics. Based on these results, it may be concluded that clozapine directly affects pancreatic insulin secretion (6).
The study performed by Chae and Kang compared the effect of clozapineand the classical antipsychotic haloperidol on glucose metabolism through oral glucose tolerance test (OGTT). Following 8 weeks of therapy, 35% patients who were administered clozapine had impaired glucose tolerance, while no such impairment was observed in patients onhaloperidol (7).
Henderson et al. included 82 patients affected by schizophrenia or schizoaffective psychosis in a 5-year follow-up. Diabetes developed in 37% of patients during the 5-year monitoring. Their mean age was 36 years and mean body mass index was 27. Results of the study showed that diabetes development correlated to patients’ age, while no correlation was recorded for body weight, changes in body weight, and changes in daily clozapine doses. Family history was positive for type 2 diabetes in only two patients (8). It has not been elucidated yet if the type and severity of mental disorder affects the risk for diabetes development in patients on clozapine (9).
Olanzapine
In a study published in year 2000, hyperinsulinemia was established in 71% of patients with schizophrenia or related types of psychoses who were on olanzepine therapy for 6 months on average. Hyperglycemia was also confirmed in three of these patients, and indicated the onset of diabetes (10).
Another study showed the development of hyperinsulinemia with or without hyperglycemia in 44% of patients who were on olanzepine therapy for the period of 25 months (11).
A prospective study involving a 14-week period demonstrated the development of intermittent or permanently present hyperglycemia in 11% of olanzepine-treated patients. All included patients formerly reacted poorly to therapy by other antipsychotics (12).
Quetiapine
An open, non-randomized retrospective study investigated changes in body weight and diabetic status of 65 patients who were on clozapine as monotherapy for 6 months, and then were administered combined clozapine and quetiapine therapy for ten months. The result was substantial reduction in body weight in all patients and better control of glycemia in 20% of patients who previously developed diabetes while on clozapine monotherapy (13). These results indicate that reduction in clozapine dose leads to reduction in body weight that is caused by this drug, and that quetiapine has no significant effect on body weight change and glucose metabolism (13).
Risperidone
Apart from several comparative studies, only two case reports of patients on risperidone have been published which showed that this drug did not lead to any complications related to regulation of glycemia in psychotic patients with comorbid diabetes (14,15).
Presently, literature provides no data on side-effects of amilsulpride,aripiprazole, ziprasidone and zotepine that are related to disturbance in glucose and insulin homeostasis (9).
Case reports
During past 15 years, numerous case reports were published on patients who developed diabetes, diabetic ketoacidosis, and hyperosmolar diabetic coma after introduction of atypical antipsychotics in therapy, or who experienced impaired glycemic control in preexisting diabetes. Data vary depending on the source, yet most of these studies are related to olanzapine and clozapine, then to risperidone, with only several studies published for quetiapine and one for ziprasidone, while no case reports have been published for zotepine, aripiprazole and amilsulpride (9). There have also been reports of patient death due to hyperosmolar diabetic coma or metabolic acidosis or ketosis during olanzapine and clozapine therapy (16-20). Koller et al. presented reports on patients with newly diagnosed diabetes, i.e. 242 patients on clozapine therapy, 225 on olanzapine therapy, and 78 patients on risperidone therapy. The numbers of patients who developed metabolic acidosis or ketosis were 80, 100, and 26, respectively (21-23). Authors reported cases of death during hyperglycemic episodes for 25 patients on clozapine, 25 patients on olanzapine, and 4 patients on risperidone (21-23).
Ananth et al. published a review demonstrating recovery after therapy discontinuation in 71% of patients with diabetes or diabetic ketoacidosis caused by therapy with atypical antipsychotics (24). Based on these observations, it may be concluded that metabolic side-effects withdraw after therapy discontinuationin most, yet not in all patients.
Possible mechanisms by which atypical antipsychotics induce insulin resistance and diabetes
The factors that may cause tissue resistance to insulin are aging, family history of type 2 diabetes, ethnic origin, obesity, smoking, and lack of physical activity (3,4).
Atypical antipsychotics cause body weight gain (25-29), sedate patients and thereby act indirectly to reduce body weight, and are thus considered to enhance insulin resistance which results in hyperlipidemia and increased risk for the development of type 2 diabetes and cardiovascular diseases (30,31).
Another mechanism by which atypical antipsychotics enhance diabetes risk is a direct effect on glucose and insulin balance; actually, clozapine and olanzapine inducehyperinsulinemia and hyperlipidemia (6,10,11,32). Hyperinsulinemia and hyperlipidemia themselves augmentinsulin resistance (31) and thereby also the risk for the development of type 2 diabetes.
With regard to drug-induced diabetes, age was shown to be a risk factor for its development (8), while smoking and lack of physical activity most probably do not represent significant risk factors for this type of diabetes.
Unlike in type 2 diabetes, family history of the disease and body weight gain are not significant risk factors for the development of drug-induced diabetes (8,33).
Possible correlation of schizophrenia and diabetes
The prevalence of type 2 diabetes is higher in patients affected by schizophrenia than in thegeneral population (34). No research has been done so far to account for this difference.
Possible reason is that important risk factors for the development of type 2 diabetes, like obesity, smoking and lack of physical activity, are more frequently observed in schizophrenic patients than in thegeneral population (35-37).
Data are available to demonstrate that schizophrenia itself as a disease is related to impaired glucose tolerance and diabetes, yet the nature of the risk that a type or severity of a psychotic disorder pose for the development of type 2- or drug-induced diabetes is unknown (9).
Atypical antipsychotics and serum lipids
The studies dealing with the effect of atypical antipsychotics on serum lipids are relatively few. Studies are described in the following paragraphs according to the antipsychotics involved (clozapine, olanzapine, risperidone, quetiapine, ziprasidone, aripiprasole).
Clozapine
Based on the hitherto published data, clozapine induces an increase in the level of triglycerides. Analysis of data from the Iowa Medicaid Program (38) compared the occurrence of hyperlipidemia in clozapine-treated patients and those treated by conventional antipsychotics. Overall, the difference in hyperlipidemia incidence between the two patient groups was not statistically significant. However, after patient distribution according to age groups, the incidence of hyperlipidemia was shown in clozapine-treated patients aged 20-34 years to be substantially higher compared to the same age group on therapy with conventional antipsychotics.
Another study showed the effect of clozapine on the change in serum lipid level during a 12-month follow-up period (39). At the end of the study, the number of patients with hypertriglyceridemia was significantly higher (19 patients) in relation to the study onset (7 patients). Also, a substantial body weight gain was observed in hypertriglyceridemic patients.
Effect of clozapine on cholesterol level has not yet been sufficiently elucidated. Actually, from all studies dealing with this topic so far, elevated cholesterol levels were observed in clozapine-treated patients in only two studies (39,40), while other studies stated no significant clozapine effect on cholesterol (41,42,43).
Risperidone
The results obtained by analyzing data on more than 18.000 patients from UK General Practice Research Database (UK GPRD) showed that risperidone was to no significant extent associated with hyperlipidemia (44).
A retrospective study conducted by Meyer (45) examined changes in serum levels of cholesterol and triglycerides in patients on risperidone during a 12-month period. The results demonstrated that this antipsychotic did not have any significant effect on serum cholesterol concentrations but caused concurrent increase in triglyceride level. Risperidone was also associated with body weight gain which positively correlates with serum lipid levels.
Other studies that may be found in the literature also confirm that risperidone has no substantial effect on changes in serum cholesterol levels, while data on its effect on triglyceride concentrations are insufficiently consistent. In fact, only one study reported a rise in triglyceride levels that was not statistically significant (45).
Olanzapine
Analysis of the data from the UK General Practice Reserach Database, UK GPRD, demonstrated that olanzapine administration was associated with elevated risk for the development of hyperlipidemia(44).
A similar result was also obtained in Meyer’s retrospective study (45) which demonstrated a significant increase in the levels of both triglycerides and cholesterol of olanzapine-treated patients. This drug also caused body weight gain which was, however, not substantially correlated to changes in cholesterol and triglyceride concentrations. It is also interesting that highest triglyceride levelswere registered during the first year of olanzapine administration.
Several other studies also confirmed the significant effect of olanzapine on the rise in the serum levels of lipids, i.e. triglycerides (46-49), total cholesterol (47,18) and LDL-cholestol (18,50), and on HDL-cholesterol decline (51).
Quetiapine
The studies investigating quetiapine effecton serum lipids have been relatively scarce and with rather inconsistent results.
A small-scale study conducted by Wirshing et al. (41) showed that quetiapine therapy had a favorable effect on triglyceride and LDL-cholesterol levels, i.e. it reduced them, while only minimum changes occurred in HDL-cholesterol concentrations.
In contrast, the laboratory results gathered in one of the clinical studies dealing with quetiapine have revealed elevated total cholesterol and triglycerides.
Ziprasidone
Results of a study that investigated the effect of ziprasidone on serum lipids in patients with mental retardation and behavioral disorders showed favorable ziprasidone action on lowering cholesterol and triglyceride concentrations (52).
Five short-term clinical studies that examined effects of ziprasidone in schizophrenic patients showed that ziprasidone therapy led to substantial reduction in cholesterol and triglyceride levels (53).
Presently, the number of studies and the amount of data on the effect of ziprasidone on serum lipids are insufficient to allow us to draw relevant conclusions. Actually, the highest amount of data is evidence of the fact that ziprasidone has no significant impacton cholesterol and triglyceride concentrations, or, as stated above, that it may have favorable effect on lipid concentrations in patients with previously elevated concentrations of these lipids.
Aripiprazole
Currently available information indicates that aripiprasole could have a favorable effect on serum lipid levels. Actually, changes in cholesterol and triglyceride levels during aripiprasole therapy were shown to be consistent with changes during placebo therapy (54); in some cases, aripiprasole administration led to a decrease in serum lipid levels which was affected by administration of other antipsychotics (55).
Atypical antipsychotics and leptin
Increased serum levels of leptin change the sensitivity of peripheral tissues to insulin, and hyperleptinemia is considered a potential link between obesity and insulin resistance (56-58).
Very little data are available on the effect of atypical antipsychotics on leptin.
It has been established that clozapine and olanzapine significantly elevate the serum level of leptin which is only slightly increased by quetiapine and minimally by risperidone.
Although past studies and other data have not offered sufficiently clear conclusions on the impact of atypical antipsychotics on metabolic parameters, atypical antipsychotics may be ranked according to the relative risk for the development of side-effects. These facts are important to keep in mind when deciding for an antipsychotic therapy which, as a rule, is a long-termone.
Clozapine and olanzapine carry the highest risk for the development of disturbance of glucose and insuline balance and of lipid and leptin levels. Quetiapine is associated with a moderate risk, risperidone with low risk, while ziprasidone and aripiprazole carrythe lowest risk.
The question arises with regard to how to prescribe atypical antipsychotics while actually being aware of possible occurrence of metabolic side-effects. We state recommendations for the monitoring of metabolic status of patients treated by antipsychotics:
1. Antropometric measurements should be performed initially when introducing antipsychotic therapy (height, weight, waist and hip measurement). Glucose, glycosylated hemoglobin, cholesterol, triglycerides, HDL-cholesterol, LDL-cholesterol, lipoprotein (a), apolipoprotein A1, Apolipoprotein B, HDL2-cholesterol and HDL3-cholesterol should be determined in serum.
2. Monthly check-ups of anthropometric measures should be performed during the first three months of antipsychotic therapy, together with the determination of glucose, cholesterol, triglycerides, HDL-cholesterol, LDL-cholesterol in serum.
3. During continued treatment with antipsychotics, anthropometric check-ups should be performed every six months, as well as determination of glucose, glycolysated hemoglobin, cholesterol, triglycerides, HDL-cholesterol, and LDL-cholesterol in serum.
4. Other initially performed tests should be repeated only if some metabolic parameters have been changed on comparison to initial examination.
5. Patients with diabetes and/or hyperlipidemia diagnosed prior to therapy should be treated by atypical antipsychotics that have lesser impact on metabolic parameters.
6. In patients who develop hyperinsulinemia, hyperlipidemia or type 2 diabetes during therapy, the medication administered should be discontinued and therapy should be instituted of another antipsychotic whose administration carries lower risk for the development of side-effects. If the newly introduced antipsychotic is not sufficiently effective in treating a psychotic disorder, the therapy is continued with the lowest effective dose of the previously used medication.
7. Continual monitoring of body weight and metabolic parameters is needed. Patient history should be examined for possible diabetes symptoms like polydypsia, polyuria, body weight loss, etc.
8. The drugs that induce high body weight gain in patients should be replaced by other drugs that are characterized by lesser expression of this undesirable effect.
9. Patients should be encouraged to lifestyle changes: smoking cessation and increased physical activity in order to diminish the risk for the development of type 2 diabetes.
Metabolic side-effects of atypical antipsychotics are increasingly manifested and may have negative influence on mental disease treatment on one hand, and physical welfare of a patient on the other. It is therefore important to keep in mind the possibility of occurrence of metabolic side-effects when prescribing these drugs, promptly recognize their effects, and continue with the treatment in a reasonable manner.
References
1. Hiles BW. Hyperglycemia and glucosuria following chlorpromazine therapy. JAMA 1956;162:1651.
2. Cooperberg AA, Eidlow S. Haemolytic anaemia, jaundice and diabetes mellitus following chlorpromazine therapy. CMAJ 1956;75:746-9.
3. Zimmet PZ. Kelly West Lecture 1991. Challenges in diabetes epidemiology: from West to the rest. Diabetes Care 1992;15:232-52.
4. Olefsky JM. Insulin resistance. In: Porte D, Sherwin RS, ed. Diabetes mellitus. 5th ed. Stamford (CT): Aplleton&Lange, 1997:513-52.
5. Comi RJ. Drug-induced diabetes mellitus. In: LeRoith D, Taylor SI, Olefsky JM, ed. Diabetes mellitus: a fundamental and clinical text. Philadelphia (PA): Lippincott Williams&Wilkins, 2000:528-8.
6. Melkersson KI, Hulting AL, Brismar KE. Different influences of classical antipsychotics and clozapine on glucose-insulin homeostasis in patients with schizophrenia or related psychoses. J Clin Psych 1999;60:783-91.
7. Chae B-J, Kang B-J. The effect of clozapine on blood glucose metabolism. Hum Psychopharmacol Clin Exp 2001;16:265-71.
8. Henderson DC, Cagliero E, Gray C, Nasrallah RA, Hayden DL, Schoenfeld DA, et al. Clozapine, diabetes mellitus, weight gain, and lipid abnormalities: a five year naturalistic study. Am J Psychiatry 2000;157:975-81.
9. Melkersson K, Dahl M. Adverse metabolic effects associated with atypical antipsychotics. Drugs 2004:64(7):701-23.
10. Melkerrson KI, Hulting AL, Brismar KE. Elevated levels of insulin, leptin, and blood lipids in olanzapine-treated patients with schizophrenia or related psychoses. J Clin Psychiatry 2000:61:742-9.
11. Melkerrson KI, Dahl ML. Relationship between levels of insulin or triglycerides and serum concentrations of atypical antipsychotics clozapine and olanzapine in patients on treatment with therapeutic doses. Psychopharmacology 2003;170:157-66.
12. Lindenmayer JP, Smith RC, Singh A, Parker B, Chou E, Kotsaftis A. Hyperglycemia in patients with schizophrenia who are treated with olanzapine. J Clin Psychopharmacology 2001;21:351-3.
13. Reinstein MJ, Sirotovskaya LA, Jones LE, Mohan S, Chasanov MA. Effect od clozapine-quetiapine combination therapy on weight and glycaemic control: preliminary findings. Clin Drug Invest 1999;18:99-104.
14. Melamed Y, Mazeh D, Elizur A. Risperidone treatment for a patient suffering from schizophrenia and IDDM. Can J Psychiatry 1998;43:956.
15. Madhusoodanan S, Brenner R, Araujo L, Abaza A. Efficacy of risperidone treatment for psychoses associated with schizophrenia, schizoaffective disorder, bipolar disorder, or senile dementia in 11 geriatric patients: a case series. J Clin Psychiatry 1995;56:514-8.
16. George K, Alberti MM. Diabetic acidosis, hyperosmolar coma, and lactic acidosis. In: Becker KL, ed. Principles and practice of endocrinology and metabolism. 3rd ed. Philadelphia (PA): Lippincott Williams & Wilkins, 2001:1438-51.
17. Von Hayek D, Hüttl V, Reiss J, Schweiger HD, Füessl HS. Hyperglycaemia and ketoacidosis on olanzapine (in German). Nervenarzt 1999;70: 836-7.
18. Zajecka JM, Weisler R, Sachs G, Swann AC, Wozniak P, Sommerville KW. A comparison of efficacy, safety and tolerability of divalproex sodium and olanzapine in the treatment of bipolar disorder. J Clin Psychiatry 2002;63:1148-55.
19. Casey DE. Dyslipidemia and atypical antipsychotic drugs. The Journal of Clinical Psychiatry, 2004;65(18):237-35.
20. Meatherall R, Younes J. Fatality from olanzapine induced hyperglycaemia. J Forensic Sci 2002;47:893-6.
21. Koren W, Kreis Y, Duchowiczny K, Prince T, Sancovici S, Sidi Y, et al. Lactic acidosis and fatal myocardial failure due to clozapine. Ann Pharmacother 1997;31:168-70.
22. Koller E, Schneider B, Bennett K, Dubitsky G. Clozapine-associated diabetes. Am J Med 2001;111:716-23.
23. Koller EA, Doraiswamy PM. Olanzapine-associated diabetes mellitus. Pharmacotherapy 2002;22:841-52.
24. Koller EA, Cross JT, Doraiswamy PM, Schneider BS. Risperidone-associated diabetes mellitus: a pharmacovigilance study. Pharmacotherapy 2003; 23:735-44.
25. Ananth J, Venkatesh R, Burgoyne K, Gunatilake S. Atypical antipsychotic drug use and diabetes. Psychoter Psychosom 2002;71:244-54.
26. Taylor DM, McAskill R. Atypical antipsychotics and weight gain: a systematic review. Acta Psychiatr Scand 2000;101:416-32.
27. Russell JM, Mackel JA. Bodyweight gain associated with atypical antipsychotics: epidemiology and therapeutic implications. CNS Drugs 2001;15:537-51.
28. Sussman N. Review of atypical antipsychotics and weight gain. J. Clin Psych 2001;62(23 Suppl.):5-12.
29. Wetterling T. Bodyweight gain with atypical antipsychotics: a comparative review. Drug Saf 2001;24:59-73.
30. Nasrallah H. A review of the effect of atypical antipsychotics on weight. Psychoneuroendocrinology 2003;28:83-96.
31. Harris M, Cahill G. Classification and diagnosis of diabetes mellitus and other categories of glucose intolerance. Diabetes 1979;28:1039-57.
32. Reaven GM. Role of insulin resistance in human disease. Diabetes 1988;37:1595-607.
33. Meyer JM. Novel antipsychotics and severe hyperlipidemia. J Clin Psychopharmacol 2001;21:369-74.
34. Melkersson K, Hulting A-L, Brismar K. Reply: body weight gain, insulin and leptin in olanzapine-treated patients (letter). J Clin Psychiatry 2001; 62:903-4.
35. Mukherjee S, Decina P, Bocola V, Saraceni F, Scapicchio PL. Diabetes mellitus in schizophrenic patients. Comphr Psychiatry 1996;37:68-73.
36. Gopalaswamy AK, Morgan R. Too many chronic mentally disabled patients are too fat. Acta Psychiatr Scand 1985;72:254-8.
37. Allison DB, Fontaine KR, Heo M, Mentore JL, Cappelleri JC, Chandler LP, et al. The distribution of body-mass index among individuals with and without schizophrenia. J Clin Psychiatry 1999;60:215-20.
38. Kelly C, McCreadie RG. Smoking habits, current symptoms, and premorbid characteristic of schizophrenic patients in Nithsdale, Scotland. Am J Psychiatry 1999;156:1751-7.
39. Lund BC, Perry PJ, Brooks JM, Arndt S. Clozapine use in patients with schizophrenia and the risk of diabetes, hyperlipidemia, and hypertension: a claim-based approach. Arch Gen Psychiatry 2001;58:1172-6.
40. Baymiller SP, Ball P, McMahon RP, Buchanan RW. Serum glucose and lipid changes during the course of clozapine treatment: the effect of concurrent beta-adrenergic antagonist treatment. Schizophrenic Res 2003;59:49-57.
41. Lindenmayer JP, Czobor P, Volavka J, Citrome L, Sheitman B, McEvoy JP, et al. Changes in glucose and cholesterol levels in patients with schizophrenia treated with typical or atypical antipsychotics. Am J Psychiatry 2003;160:290-6.
42. Wirshing DA, Boyd JA, Meng LR, Ballon JS, Marder SR, Wirshing WC. The effects of novel antipsychotics on glucose and lipid levels. J Clin Psychiatry 1999; 156:1270-2.
43. Spivak B, Lamschtein C, Talmon Y, Guy N, Mester R, Feinberg I, et al. The impact of clozapine treatment on serum lipids in chronic schizophrenic patients. Clin Neuropharmacol 1999;22:98-101.
44. Spivak B, Roitman S, Vered Y, Mester R, Graff E, Talmon Y, et al. Diminished suicidal and aggressive behavior, high plasma norepinephrine levels, and serum triglyceride levels in chronic neuroleptic-resistant schizophrenic patients maintained on clozapine. Clin Neuropharmacol 1998;21:245-50.
45. Koro CE, Fedder DO, L’Italien GJ, Weiss S, Magder LS, Kreyenbuhl J, et al. An assessment of the independent effects of olanzapine and risperidone exposure on the risk of hyperlipidemia in schizophrenic patients. Arch Gen Psychiatry 2002; 59:1021-6.
46. Meyer JM. A retrospective comparison of weight, lipid and glucose changes between risperidone and olanzapine treated inpatients: metabolic outcomes after 1 year. J Clin Psychiatry 2002;63:425-3.
47. Dimelis D, Garyfallos G, Kiouniakis F et al. Olanzapine vs. risperidone: impact on weight gain and blood lipids: is there any relationship with antipsychotic efficacy? Eur Neuropsychopharmacol 2002; 12(suppl 3):S328.
48. Conley RR, Mahmoud R. A randomized double-blind study of schizophrenia or schizoaffective disorder. Am J Psychiatry 2001;158:756-74.
49. Osser DN, Najarian DM, Dufresne RL. Olanzapine increases weight and serum triglyceride levels. J Clin Psychiatry 1999;60:767-70.
50. Ishigooka J, Murasaki M, Miura S, and the Olanzapine Early-Phase II Study Group. Efficacy and safety of olanzapine, an atypical antipsychotic, in patients with schizophrenia:results of open-label multicenter study in Japan. Psychiatry Clin Neurosci 2001;55:353-63.
51. Simpson GM, Weiden P, Piggot T, et al. Ziprasidone vs olanzapine in schizophrenia: 6-month continuation study. Eur Neuropsychopharmacol 2002;12(suppl 3):S311.
52. Kurt E, Oral ET. Antipsychotics and glucose, insulin, lipids, prolactin, uric and metabolism in schizophrenia. Eur Neuropsychopharmacol 2002;12 (suppl 3):S276.
53. Cohen S, Fitzgerald B, Okos A, Khan S, Khan A. Weight, lipids, glucose and behavioral measures with ziprasidone treatment in a population with mental retardation. J Clin Psychiatry 2003;64:60-2.
54. Romano S, Cutler N, Weiden PJ, et al. Ziprasidone’s effect on weight and lipids in patients with schizophrenia. Int J Neuropsychopharmacol 2002;5:S171.
55. Marder SR, McQuade RD, Stock E, Kaplita S, Marcus R, Safferman AZ, et al. Aripiprazole in the treatment of schizophrenia: safety and tolerability in short-term, placebo-controlled trials. Neuropsychopharmacology 1999;20:491-505.
56. Segal KR, Landt M, Klein S. Relationship between insulin sensitivity and plasma leptin concentrations in lean and obese men. Diabetes 1996;45:988-91.
57. Haffner SM, Miettinen H, Mykkanen L, Karhapaa P, Rainwater DL, Laakso M. Leptin concentrations and insulin sensivity in normoglycaemic men. Int J Obes Relat Metab Disord 1997;21:393-9.
58. Girard J. Is leptin the link between obesity and insulin resistance? Diabetes Metab 1997;23:16-24.