Introduction
Activated partial thromboplastin time (APTT) is a screening test used to assess the intrinsic pathway of coagulation, to monitor heparin treatment and to detect hemorrhagic disorders (1). Partial thromboplastin consists only of phospholipids to which a certain surface activator with negative surface charge (silicon dioxide, kaolin, ellagic acid, cephalin, etc.) has been added to activate the intrinsic pathway. The time to the fibrin clot formation is measured in seconds.
A number of different reagents for APTT determination are commercially available. They differ according to the partial thromboplastin sensitivity and choice of surface activator. The sensitivity of partial thromboplastin depends on its origin and concentration. The phospholipids comprising partial thromboplastin may be isolated from the placental or brain tissue, or may be of plant origin. The reagents also differ according to the type of surface activator: silicon dioxide (Pathromtin SL), ellagic acid (Dade Actin FS) and polyphenols (STA Cephascreen). Depending on the reagent composition, manufacturers suggest different reference ranges. The suggested values for Pathromtin SL are 25.9–36.6 seconds, for Dade Actin FS 23.0–31.9 seconds and for STA Cephascreen 24–35 seconds. It is recommended that each laboratory determine its own reference range of APTT in dependence of the reagent used (1).
Different composition of reagents results in variable APTT values measured in the same plasma sample, especially in heparinized plasma, because heparin binds to antithrombin III, thus precipitating its inhibitory action upon activated factors in the intrinsic pathway (7).
This observation stimulated us to compare the APTT values obtained by Pathromtin SL and Dade Actin FS reagents on Behring Coagulation System analyzer, and by STA Cephascreen reagent on STA Compact analyzer.
APTT ratio has been introduced as a unit to express the measured values in order to reduce differences in APTT results due to different reagents. APTT ratio is a quotient between the number of seconds of the APTT measured and the mean reference range of the reagent used. According to recommendations issued by the Croatian Chamber of Medical Biochemists, the reference range for individuals older than six months is 0.8–1.2 (8).
In this study, we compared APTT values in seconds because the values of commercial control materials are declared in seconds and analyzer’s units are also expressed seconds.
Materials and methods
The study included 114 fresh citrated plasma samples from 68 subjects without heparin therapy, with APTT values within the reference range of Pathromtin SL reagent (26–37 s), and from 46 subjects known to receive therapy with low molecular weight heparin. APTT values were determined in parallel by use of Pathromtin SL and Dade Actin FS reagents on a Behring Coagulation System analyzer (Dade Behring GmbH, Marburg, Germany) and by use of STA Cephascreen reagent on an STA Compact analyzer (Roche Diagnostics GmbH, Mannheim, Germany). Samples were analyzed by the coagulometry method with all three reagents and on both analyzers immediately upon the receipt at coagulation laboratory during March 2006.
APTT values were determined continuously for 31 days in the Bio-Rad Lypocheck Coagulation Control samples (Level 1, 2 and 3) by use of the mentioned reagents.
Statistical analysis
Correlation of the APTT results was performed. The level of significance was set at P < 0.01. Differences between correlation coefficients were tested for the statistical significance.
Passing-Bablok regression was used on APTT value comparison, including the Cusum test for linearity. Statistical analysis including descriptive statistics was performed by use of the MedCalc 9.2.0.0 software (MedCalc, Mariakerke, Belgium).
Results
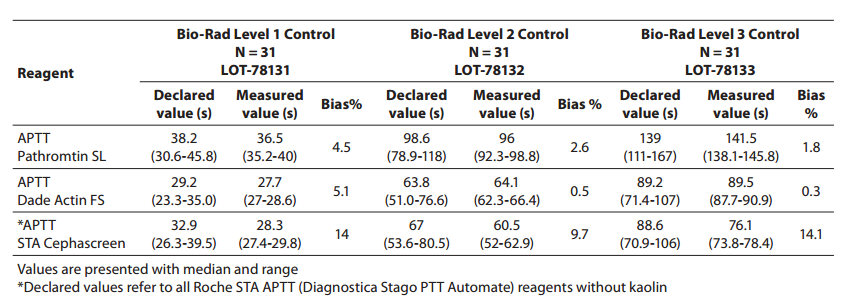
The values recorded in Bio-Rad controls were within the range reported by the manufacturer at all three control levels (Level 1, 2 and 3). The APTT values obtained in Bio-Rad controls (Level 1, 2 and 3), the values declared by the manufacturer and the bias are presented in Table 1. The largest bias of 14% was recorded with STA Cephascreen reagent.
Table 1. Comparison of declared APTT values of Bio-Rad controls and measured values for all reagents used
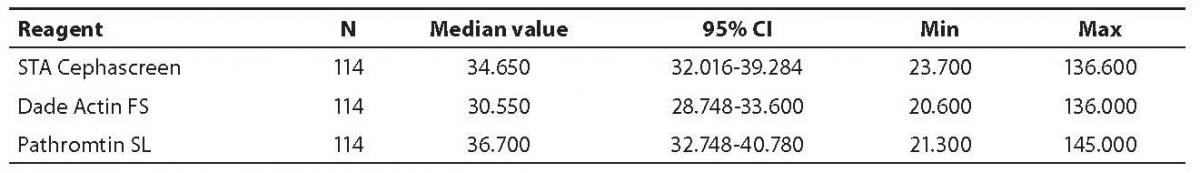
The median of APTT values measured in 114 plasma samples was from 30.55 to 36.70 s for all three reagents used. Complete descriptive statistics is shown in Table 2.
Table 2. Descriptive statistics for APTT values obtained with the three reagents
Correlation of APTT values obtained by use of different reagents yielded the following correlation coefficients: Pathromtin SL vs.Dade Actin FS 0.9485; Dade Actin FS vs. STA Cephascreen 0.9353; and Pathromtin SL vs. Cephascreen 0.9072 (Table 3).
Table 3. Correlation coefficients for all three reagent combinations in all study samples
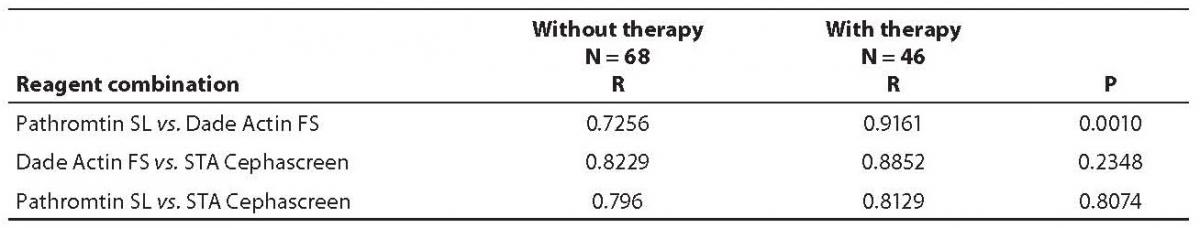
The study population of 114 subjects was categorized into two groups: without heparin or heparin derivative therapy (N = 68) and the group on therapy with low molecular weight heparin (N = 46). Correlation coefficients and presence of statistically significant between-group differences were determined in these two groups (Table 4).
Table 4. Correlation coefficients of APTT results in groups without and with therapy determined with all three reagents used
A statistically significant difference in correlation coefficients was recorded between the results obtained by use of Pathromtin SL and Dade Actin FS reagents in the groups with and without heparin therapy (P = 0.001). Passing-Bablok regression was performed for each group in separate.
Other reagent combinations (Dade Actin FS vs. STA Cephascreen and Pathromtin SL vs. STA Cephascreen) yielded no statistically significant difference in correlation coefficients, thus being considered as a homogeneous group.
The Cusum linearity test performed on Passing-Bablok regression showed that there was no significant deviation from linearity for any of the above reagent combinations (P > 0.10).
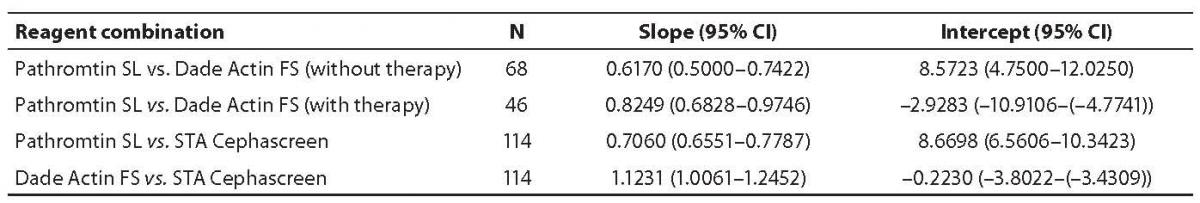
The slope and y-axis intercept as well as 95% confidence intervals yielded by Passing-Bablok regression for various reagent combinations are presented in Table 5.
Table 5. Passing Bablok regression data – slope and intercept with 95% confidence interval
All data in the Results section that were presented in seconds were re-calculated as ratio depending on the reference range for each reagent used. These data are not shown because the statistical features, values of tests and their interpretation correspond to those in seconds. Considering that the values of commercial control materials are declared in seconds, as mentioned in the Introduction, we decided to express all study results in seconds in order to sustain consistency of the result presentation.
Discussion
The aim of the study was to verify our daily routine experience with comparability of different reagents used on APTT determination. Pathromtin SL, Dade Actin FS and STA Cephascreen showed variable sensitivity according to the commercial control samples, Bio-Rad Level 1, 2 and 3. All three reagents performed well in terms of preset values declared by the manufacturer but differed between each other. The largest bias from the declared values was observed with STA Cephascreen reagent. It should be noted that the manufacturer of the commercial controls divides Roche STA APTT (Diagnostica Stago PTT Automate) reagents into groups with or without kaolin, meaning there are no specific values for STA Cephascreen reagent. According to this, STA Cephascreen reagent is in the “without kaolin” group.
It should be noted that correlation coefficients yielded high correlation for all the reagent combinations tested, with the condition of linearity met in all of these combinations. However, additional analysis by use of Passing-Bablok regression revealed some discrepancies. Comparison of Dade Actin FS and Pathromtin SL reagents indicated that, in spite of the high correlation, patients should not be tested as a homogeneous group if receiving low molecular weight heparin therapy.
A significantly lower correlation coefficient was obtained in the group without heparin therapy than in the group of patients administered heparin therapy, although the result difference recorded prior to group categorization seemed to be by far greater relative to high APTT values.
The slope 95% confidence interval did not include 1 in either group, whereas the y-axis intercept 95% confidence interval in the group without therapy did not include 0 for difference from the group with heparin therapy, where it included 0. Also, the slope was not comparable in the two groups, thus additionally indicating low compatibility level. These data show that there was no proportional result deviation, i.e. the level of comparability was rather low.
The fact that the results obtained by use of Pathromtin SL and Dade Actin FS reagents had to be tested according to categories of patients with or without heparin therapy points to the relatively poor comparability of the two reagents. The slope and y-axis intercept obtained by Passing-Bablok regression for Pathromtin SL and STA Cephascreen did not include 1 and 0, respectively, also pointing to discrepancy of the results showing variable linearity. Dade Actin FS and STA Cephascreen were the only combination meeting the conditions of Passing-Bablok regression, i.e. the slope was very close to 1, while the y-axis intercept was small. Thus, the results obtained by Dade Actin FS and STA Cephascreen reagents were quite compatible and followed the same linearity pattern.
Accordingly, the highest correlation was observed between the results of APTT measurement by use of Dade Actin FS and STA Cephascreen reagents, whereas the results obtained by use of Pathromtin SL and Dade Actin FS yielded lowest correlation. Greater result deviation was observed in the group of samples without heparin therapy, which the manufacturer had probably tried to correct by proposing different reference ranges.
Thus, although very good correlations were obtained for all three reagents, the APTT values determined by different reagents showed differences that could not be disregarded.
It should be noted that Pathromtin SL and Dade Actin FS reagents come from the same manufacturer and the phospholipids found in the two reagents are of plant origin, whereas the phospholipid moiety of the STA Cephascreen reagent is of animal origin (cephalin from rabbit brain tissue). As the reagents differ substantially according to the type of activator surfaces, i.e. silicon dioxide (Pathromtin SL), ellagic acid (Dade Actin FS) and polyphenols (STA Cephascreen), the type of activator surface may play a decisive role in the particular reagent sensitivity on APTT determination.
All these data suggest that it is of great importance to follow APTT results of each individual patient using the same reagents on the same analyzer.