Introduction
Detection of antinuclear antibodies (ANA) is extensively used for establishing a diagnosis in patients with clinical features suggestive of non-organ specific (systemic) or organ-specific autoimmune disorders. Among the systemic autoimmune diseases that are characterized by the presence of ANA there are systemic lupus erythematosus (SLE), Sjögren's syndrome (SS), systemic sclerosis (SSc), inflammatory myositis (IM), mixed connective tissue disorder (MCTD), rheumatoid arthritis (RA) and drug induced SLE. The diagnosis of these diseases depends on the identification of disease-associated clinical symptoms and signs, and is associated with the detection of autoantibodies directed against nuclear or cytoplasmic antigens (1). Autoimmunity laboratories analyze and measure an increasing number of autoantibodies, employing a broad spectrum of techniques and methods. The main characteristic of the autoimmunity laboratory, and indeed the one that differentiates it from other laboratories that use immnunoassays as the basic technique, is that it determines antibodies (autoantibodies) and not antigens (2).
The most common method for the identification of positive patient's sera for ANA is indirect immunofluorescence (IIF), but this method gives high positive (false) responses among healthy individuals and variability in results between laboratories (3). Detection of ANAs by IIF is a microscopic technique, in which reader agreement and reliability are of great importance. IIF provides an approximation to the identification of possible autoantigens by means of their fluorescence patterns. Furthermore, the manual IIF procedure is relative labor-intensive. Considerable effort has been made in developing simpler automated assays for routine laboratory use. Several different forms of enzyme immunoassays have been developed which were simpler, high-throughput analyses that can be automated and standardized. (4) Of these, enzyme-linked immunosorbent assay (ELISA) based either on antigens prepared from human tumor cell line (HEp-2) nuclear extracts or from recombinant and highly purified nuclear antigens has been the most promising, but substantial differences in terms of positivity among various enzyme immunoassay methods have been described (5). The specificity of ELISAs for autoantibody measurements is strongly dependent on the quality of antigens used, and it is important that an antigen should have exactly the same sequence, conformation and post-translational modifications as the human antigen. Flow cytometry for the analysis of microsphere-based immunoassays has been developed for the simultaneous determination of different substances (6). Recently a commercially available microsphere-based fluorescent assay has been introduced for the detection of ANA. Among applications of this technology there is simultaneous measurement of several autoantibodies. The system uses polystyrene microspheres labelled internally with different ratios of two different fluorochromes. Each fluorochrome can have any of the 10 possible levels of fluorescence intensity, thereby creating a family of 100 spectrally addressed bead sets. The antigens corresponding to autoantibodies are bound to the microspheres. Each of the 100 microbeads that can be differentiated by their fluorescence carries a specific immobilized antigen for a single autoantibody. At the same time, a green laser excites the external reporter fluorescence to quantify the specific reaction related to each autoantibody (7). The AtheNA Multi-Lyte ANA test system is a multiplexed homogenous, fluorescence-based microparticle immunoassay, designed to detect and distinguish the autoantibodies present in human serum. The Luminex 100 System (Luminex Corporation, Austin, TX, USA) is a flow cytometer analyzer specifically designed for xMAP technology.
In detection of an antinuclear antibody, it is important to know when the initial IIF ANA screening on HEp-2 cells can be replaced with multiplexed bead-based immunoassay without loss of any important results. The objective of the study was to determine the performance of multiplexed bead based immunoassay (AtheNA Multi-Lyte ANA test system) relative to established, commercial IIF ANA HEp-2 and ELISA currently used in our laboratory.
Materials and methods
Patients
The study included 897 serum samples submitted to our laboratory for autoimmune testing over a period of 7 months (from June 2006 to December 2006). The sera were selected on the basis of established diagnosis or indication of autoimmune disease. All samples were divided into three groups.
One group consisted of 301 patients with established diagnosis: rheumatoid arthritis (N = 122), SLE (N = 101), Sjögren syndrom (N = 9), systemic sclerosis (N = 34) and different types of vasculitis (N = 35).
The second group consisted of 398 patients with clinical suspicion of certain autoimmune disease: rheumatoid arthritis (N = 47), SLE (N = 22), Sjögren syndrome (N = 7), systemic sclerosis (N = 23), vasculitis (N = 29) and with suspicion of autoimmune disorder (N = 270).
The third group consisted of 198 patients without diagnosis.
All positive specimens were tested for specific autoantibodies directed against anti Sjőgrens syndrome SS-A/Ro (SSA), SS-B/La, Smith antigen (Sm), ribonucleoprotein (RNP), antitopoisomerase I (Scl-70), autoantibodies directed against hystidyl-tRNA syntetase (Jo-1), autoantibodies to native double stranded DNA (dsDNA), centromere B and histone.
After blood clotting, serum was obtained by centrifugation and stored at –20 C until further analysis.
Local Ethics Committee of the Split University Hospital approved the study (13).
Methods
AtheNA Multi-Lyte ANA
A total of 897 sera were tested using multiplex technology based tests (AtheNA Multi-Lyte ANA test system, Zeus Scientific, Inc., Raritan, NJ, 08869 USA). AtheNA Multi-Lyte ANA test system was utilized for the simultaneous semi-quantitative determination of autoantibodies to nine different antigens: (SSA, SSB, Sm, RNP, Scl-70, Jo-1, dsDNA, Centromere B, Histones) and qualitative measurement of ANA. The assay is based on a microsphere-based technology. Antigen source for SSA, Sm, and histone antigens are purified from natural sources while SSB, RNP, Scl-70, Jo-1 and Cent B are human recombinant antigens. The qualitative ANA preparation is derived from the nuclei of HEp-2 cells. Sera and controls were diluted 1:21 with specimen diluent. Bead suspensions were thoroughly mixed with diluted sera and controls and incubated in a microtitre plate. If present in sera, specific autoantibodies bind to the antigen. The conjugate (goat anti-human IgG conjugate with phycoerythrin) is added after incubation and washing away of unbound material. After second incubation, the bead suspensions were thoroughly mixed and then placed on a microplate and into AtheNA reader (within 60 minutes) to read the requested results. The AtheNA Multi-Lyte instrument utilizes Luminex 100 technology (Austin, USA) which is based on flow cell fluorometry. Positivity of tests was calculated according to the manufacturer’s instructions as follows: a serum was considered positive if at least one individual parameter was > 120 AU/mL, or an internal HEp-2 extract (internal control sample) was > 120 AU/mL. A serum was considered equivocal if any test value ranged from 100 to 120 AU/mL, and negative if the corresponding fluorescence was < 100 AU/mL.
IIF ANA
A total of 897 sera were tested using convetional immunoassay including ANA HEp-2 test system (BioSystems S.A., Costa Brava 30, Barcelona, Spain). The IIF ANA uses HEp-2 cells as a substrate and goat anti-human IgG conjugated with fluorescein isothiocyanate as a conjugate. Sera were diluted 1:80 with phosphate-buffered saline and overlaid on the substrate for 30 min at room temperature. Slides were washed twice for 5 min each with phosphate-buffered saline, overlaid with conjugate and incubated for additional 30 min. Subsequently, slides were washed twice and examined with a fluorescence microscope at x 40 magnification. Patient samples were considered positive when a positive signal was obtained at dilution ≥1:80. Negative and positive controls were used to check accuracy.
ELISA
A total of 200 serum samples positive to ANA were analyzed by single-antigen ELISA for SSA, SSB, Sm, Sm/RNP, Scl-70, Jo-1, dsDNA (Hycor Biomedical Ltd, Pentlands Science Park, Bush Loan, Penincula, UK), Histone and Centromere B (DiaSorin S.p.A., Via Corescentino 13040 Sallugia, Italy). Microtitre wells were coated with antigens from the following sources: for SSA, SSB, Sm, RNP, Scl-70 and Jo-1 from bovine sources, for dsDNA the wells were coated with purified dsDNA from calf thymus, for Cent B recombinant CENT-P (bacillovirus) and for Histone from chicken bone tissue (blood stem cells). All single-antigen ELISA tests were performed at an automatic microplate analyzer (miniBOS, Biomedica Gruppe, GMBH, A-1210, Divischgasse 4, Vienna, Austria).
In principle, dilutions of controls and samples (1:100) are performed automatically by the instrument. On adding standards, diluted controls and serum to the wells, the antibodies present bind to the antigen. After incubating and washing away unbound material, a conjugate (peroxidase-labeled anti-human IgG mouse monoclonal antibody) is added. Following further incubation and washing, a substrate (tetra-methyl benzidine) is added to each well. The presence of the antigen-antibody-conjugate complex changes the substrate. The reaction is stopped by adding 0.25 M H2SO4. Absorbance is measured at 450 nm. Color intensity is proportional to the amount of the autoantibodies present in a serum sample. Test results were calculated according to manufacturers instruction and range for the single-antigen ELISA. Intra assay coefficients of variation (CV%) ranged for SSA from 2.8% to 3.1%, SSB from 5.5% to 9.1%, Sm from 5.4% to 8.6%, RNP from 2.9% to 11.2%, Scl-70 from 3.0% to 5.1%, Jo-1 from 3.6% to 6.1%, dsDNA from 3.9% to 6.9%, Centromere B from 4.8% to 8.6%, and Histone from 6.6% to 10.6%, while inter assay CVs ranged for SSA from 3.5% to 7.9%, SSB from 3.4% to 6.0%, Sm from 3.2% to 5.1%, RNP from 4.9% to 7.4%, Scl-70 from 6.7% to 9.4%, Jo-1 from 2.7% to 8.3%, dsDNA from 8.4% to 9.8%, Centromere B from 6.2% to 11.0%, and Histone from 8.3% to 12.9%.
A serum was considered positive if autoantibodies to SSA, SSB, Sm, Sm/RNP, Scl-70 and Jo-1 were > 15 U/ml, equivocal if any test value was from 10 to 15 U/mL, and negative if < 10 U/mL. Results for autoantibody to dsDNA > 60 U/mL were considered positive, the value from 40 to 60 U/mL equivocal, and < 40 U/mL negative; for Centromere B > 11 U/mL was considered positive, the value from 8 to 11 U/mL equivocal, and < 8 negative, and for Histone > 21 U/mL value was considered positive, the value from 15 to 21 U/mL equivocal, and < 15 U/mL negative.
The results obtained in this study were compared to the reference values declared by manufacturers.
Statistical analysis
Statistical analysis was performed with SPSS V 8.0 (SPSS Inc, Chicago, IL). Correlations were calculated using the Spearman rho test and P value < 0.001 was considered statistically significant. Agreement among methods was measured by calculating the kappa coefficient, P < 0.001 was considered statistically significant.
Sensitivity and specificity of the AtheNA Multi-Lyte ANA test was calculated by using the single-antigen ELISA results as the reference standard and the following definitions: true positive test result (TP), true negative test result (TN), false negative test result (FN), false positive test result (FP). Sensitivity (TP/[TP+FN] x 100) was computed as the percentage of positive test responses in sample with positive ELISA. Specificity (TN/[FP+TN] x 100) was computed as the percentage of negative tests in a sample with negative ELISA.
Results
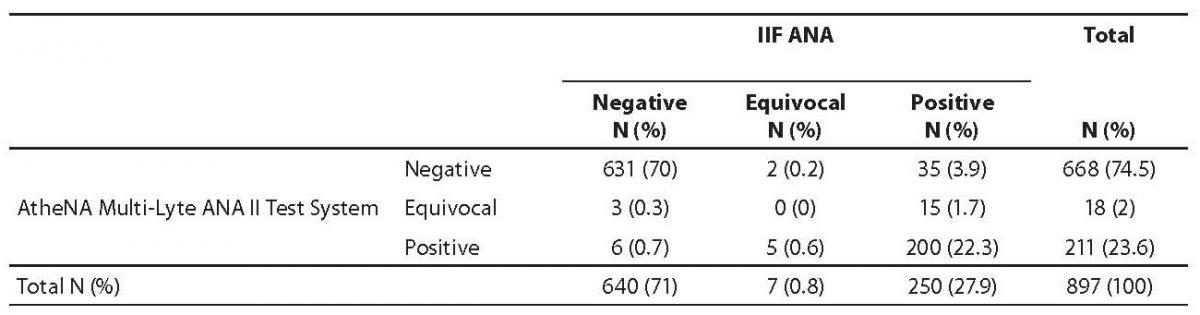
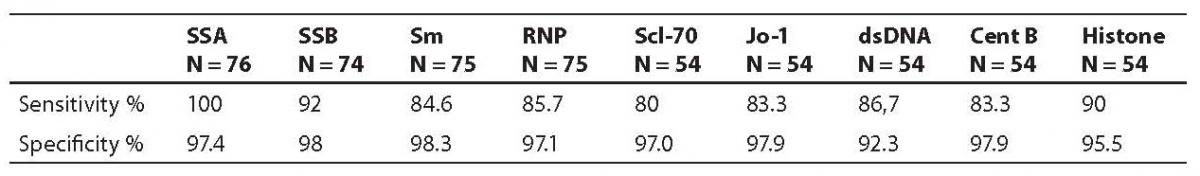
A total of 897 serum samples from patients with suspected autoimmune disorders were delivered to our laboratory for ANA testing. The data obtained by the AtheNA Multi-Lyte ANA test system (HEp 2 cell lysate only) and IIF ANA are presented in Table 1. On comparing the results obtained by the AtheNA Multi-Lyte ANA test system (expressed as qualitative results) and IIF ANA, a complete concordance was observed in 92.3% samples (70% of total samples were negative and 22.3% were positive). Fifty-nine (6.6%) samples were disconcordant, 6 (0.7%) false positive, 35 (3.9%) false negative, and 18 (2%) were equivocal. All 35 negative samples obtained with AtheNA Multi-Lyte ANA test system and IIF ANA were negative with single antigen ELISA. Fluorescence pattern observed in this samples was noted as unusually speckled (N = 7) or homogenous (N = 28). All six samples positive according to AtheNA Multi-Lyte ANA test system and IIF ANA negative were positive in single-antigen ELISA as SSA. Among 18 equivocal samples obtained with AtheNA Multi-Lyte ANA test system, three samples were IIF ANA negative (single antigen ELISA SSA were positive) and 15 samples IIF ANA positive. Of these 15 IIF ANA positive samples tested with single antigen ELISA, 6 were ds DNA positive, 4 Scl-70 positive, while 5 samples were negative. For a specific autoantibody tested using AtheNA Multi-Lyte ANA test system and single antigen ELISA, sensitivity was from 80.0% (Scl-70) to 100% (SSA) and specificity from 92.3% (dsDNA) to 98.3% (Sm), as presented in Table 2.
Table 1. Cross-tabulation of results obtained with immunofluorescence and the AtheNA Multi-Lyte ANA test System
Table 2. Sensitivity and specificity of the methods: AtheNA Multi-Lyte ANA test system and single-antigen ELISA
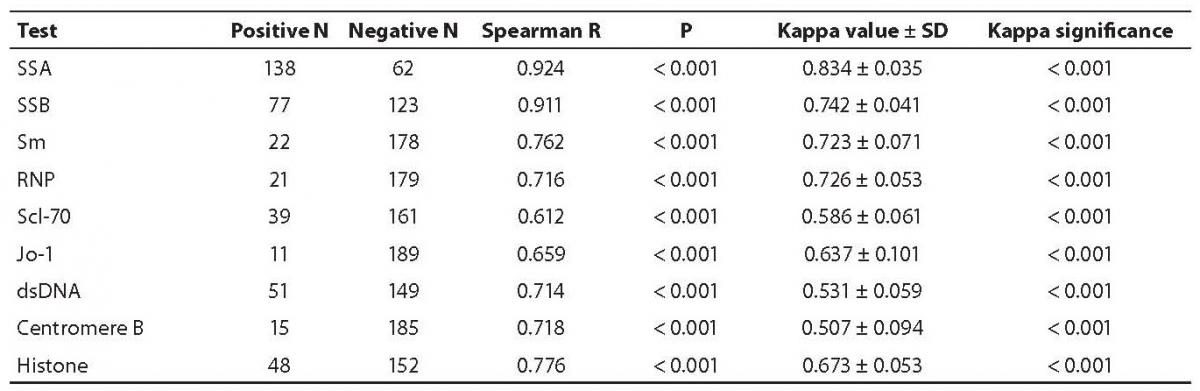
Antinuclear antibodies were positive in 200 serum samples tested with AtheNA Multi-Lyte ANA test system and IIF ANA, and expressed qualitatively. All positive sera were tested with single-antigen ELISA for the same autoantibodies obtained with AtheNA Multi-Lyte test system reacting with SSA, SSB, Sm, RNP, Scl-70, Jo-1, dsDNA, Centromere B and Histones expressed semi-quantitatively. When multiplexed-assay results were compared to single antigen ELISA for all tested parameters, a significant correlation was obtained (P < 0.001). Two methods correlated significantly for all autoantibodies (kappa value from 0.834 for SSA to 0.507 for centromere B; Table 3).
Table 3. Correlation of specific autoantibodies assayed by AtheNA Multi-Lyte ANA test system and single-antigen ELISA, and agreement between the methods
Differences in methodology between multiplexed and single ELISA assays were taken into consideration and comparison of 200 positive serum samples included combined results for single autoantibodies. When multiplexed-assay results were compared to the single antigen ELISA, there was an agreement from 90.5% to 100% (Table 4).
Table 4. Correspondence for positive antinuclear antibody in serum between AtheNA Multi-Lyte ANA test system and single-antigen ELISA
Discussion
Screening of antinuclear antibody is performed for a variety of systemic and organ specific autoimmune diseases. Different techniques have been used to develop specific tests for ANA detection including immunofluorescence, immunodiffusion, enzyme immunoassays, immunoblotting techniques and recently multiplexed bead base assays. The laboratory may have dilemmas about techniques that are relevant and reliable to detect all clinically significant autoantibodies. At the same time, the applied method must be a tool for high throughput, efficient, easy to use and inexpensive. In our study, we examined test combinations to find out the the best choice for screening antinuclear autoantibody and determination of a specific autoantibody. It is, however, only with clinical validation possible to conclude about clinical specificity and sensitivity of tests.
Indirect immunofluorescence (IIF) microscopy is a sensitive method, yet it has some limitations like substrate variations, manual performance, subjective result interpretation, low reproducibility and lack of standardization. The multiplexed bead base assay, AtheNA Multi-Lyte ANA test system, is designed for the qualitative measurement of ANA while simultaneously permitting the semi-quantitative detection of IgG class antibodies against SSA, SSB, Sm, U1-RNP, Scl-70, Jo-1, Centromere B, dsDNA and histones in human serum. These nine parameters permit the detection and quantification of the main autoantibody of systemic autoimmune disseases. This test system has been intended to become a potential replacement for conventional IIF and ELISA methods. In the present study, we compared 897 serum samples using IIF ANA and AtheNA Multi-Lyte ANA test for the qualitative measurement of ANA; the agreement between these methods was 92.3%. Among 897 tested serum samples, 35 (3.9%) samples were positive with IIF ANA test but negative with AtheNA Multi-Lyte test system (Table 1). The fluorescence pattern in these samples was observed as unusually speckled or homogeneous. IIF ANA positive samples were from a group with suspicion of autoimmune disorder and among the sera that were without diagnosis. IIF ANA testing on HEp-2 cells lacks specificity because of a considerable overlap in the clinical features of systemic autoimmune diseases, and because of the presence of various extractable nuclear antigen (ENA) antibodies. Also, IIF ANA method offers a broad screening capability and detects rarely-seen antibodies against centrioles, signal recognition particles and many other cellular targets which may be useful in clinical diagnostics (8,9). We used IIF ANA positive samples with 1:80 titer despite the recommendation by an international serology committee (10,11) because we had a great number of specimens without diagnosis or samples with possible diagnosis of an organ specific disorder. Thus we obtained high sensitivity and minor possibility to miss positive results, but also low throughput and the need for further testing with single autoantibodies. The finding of 6 (SSA) positive and 3 (SSA) equivocal results according to AtheNA Multi-Lyte test system and negative according to IIF ANA method suggested that IIF ANA HEp-2 substrate lacked sensitivity for SSA (12). Also, the samples with bordeline ANA fluorescence might contain anti SSA. Discrepant negative results present a problem because a potentially relevant antibody may be missed (14,15,16). Although screening with conventional IIF ANA HEp-2 cells misses some antigens such as SSA and Jo-1, false negative results are infrequent. Martins et al. compared single-antigen ELISAs for SSA, SSB, Sm, RNP and Scl-70 with multiplexed ENA 5 Luminex based assay. They obtained 93.6% agreement or higher for all antibodies, the sensitivity ranging from 50% for Scl-70 to100% for SSA and the specificity from 97.9% for Sm to 99.5% for SSB (17). Our results showed agreement from 90.5% for RNP to 100% for SSB, sensitivity from 80.0% for Scl-70 to 100% for SSA, and specificity from 92.3% for dsDNA to 98.3% for Sm for AtheNA Multi-Lyte ANA test system and ELISA (Table 2, Table 4). We compared AtheNA Multi-Lyte ANA test with single-antigen ELISA and found good correlation between all tested parameters (Spearman R from 0.612 to 0.924 and P < 0.001) (Table 3). A high concordance was obtained with the study that used the same technology, multiplex bead base and ELISA (17-21). Nifli et al. compared results of ELISA and AtheNA methods for SSA, SSB, Sm, RNP, Scl-70 and the correlation was significant (kappa values from 0.347 to 0.764, P < 0.001), but no correlation was established for cytoplasmic antigen Jo-1. Despite the latter, results indicate that AtheNA Multi-Lyte ANA could replace single-antigen ELISA for the measurement of specific autoantibodies (20). In the present study, two methods correlated significantly for all autoantibodies (kappa value from 0.834 for SSA to 0.507 for centromere B). Shovman et al. compared the results obtained for ANA in healthy donors, using ELISA and AtheNA Multi-Lyte ANA and demonstrated high concordance rate of 99%, with 97.7% rate found for ANA determined on requested samples provided by other laboratories. They concluded that AtheNA Multi-Lyte ANA test system is a sensitive screening method for ANA (18). The ELISA method for ANA detection could, however, miss some antigens because antigens were prepared from HEp-2 cells and were defined with a limited number of specific antigens.
Comparing single sample results for specific antigens, we found some discrepant results (Table 4). All discrepant results except three serum samples for autoantibody to Scl-70 antigen and four for dsDNA antigen were near bordeline positive or negative results. The assays possess a separate cut off value which allow comparison of obtained results. The discrepant results for three ELISA positive results for Scl-70 and four AtheNA Multi-Lyte ANA negative results for dsDNA can be explained by the source of antigens, presentation of antigens, reaction conditions of these two methods, diferences in test methodology, diferences in recombinant or purified antigens, their binding capacity and concentration in secondary antibodies and fluorescent conjugates (20, 22). The test condition including ionic strengths and cut off determination may account for differences in test results between assays. Scl-70 and dsDNA antigens were conjugated by a covalent bound to an individual set of microspheres through its surface-functional carboxyl groups. If the coupling procedure, activation, concentration of microspheres or quality of coupling antigen stabilizing solution and washing by filtration are not balanced, we can lose antigens and results will be a negative. Discrepant negative results present a problem because potentially relevant antibodies may be missed. Bordeline results must be confirmed or refuted by use of an independent method and retesting on a new serum sample during a month period. It is important to note that the AtheNA Multi-Lyte ANA test is composed of nine individual analytes while IIF ANA test is composed of numerous components present in HEp-2 cells. The diference in assays must be understood, and results from multiplex assay should always be interpreted within the clinical context.
Conclusion
The AtheNA Multi-Lyte ANA test system showed good agreement with IIF ANA and excellent results in comparison with the conventional single-antigen ELISA methods for sera with defined systemic autoimmune disease. The AtheNA Multi-Lyte ANA test supports the immediate identification of autoantibodies of clinical significance in serum samples without additional testing. For laboratory, it is more efficient, easy-to-use, and a high-throughput technique. It is important to note that the assays can be expressed in terms of agreement rates and discrepancies but, without proper clinical evaluation including population with confirmed autoimmune disease and healthy population, the value of the data obtained is rather limited.
Notes
Potential conflict of interest
None declared
References
1. Damoiseaux JGMC, Cohen Tervaert JW. From ANA to ENA: How to proceed? Autoimmunity Reviews 2006;5:10-7.
2. Gonzáles-Buitrago JM, Gonzáles C. Present and future of the autoimmunity laboratory. Clinica Chimica Acta 2006;365:50-7.
3. Hayashi N, Kawamoto T, Mukai M, Morinobu A, Koshiba M, Kondo S, et al. Detection of antinuclear antibodies by use of an enzyme immunoassay with nuclear Hep-2 cell extract and recombinant antigens: comparison with iImmunofluorescence assay in 307 patients. Clin Chem 2001;47:1649-59.
4. Fenger M, Wiik A, Høter-Madsen M, Lykkegaard JJ, Rozenfeld T, Hansen MS, et al. Detection of antinuclear antibodies by solid-phase immunoassays and immunofluorescence analysis. Clin Chem 2004;50:2141-7.
5. Tan EM, Smolen JS, McDougalJS, Butcher BT, Conn D, Dawkins R, et al. A critical evaluation of enzyme immunoassays for detection of antinuclear autoantibodies of defined specificities. Precision, sensitivity, and specificity. Arthritis Rheum 1999;42:455-64.
6. Gonzáles-Buitrago JM. Multiplexed testing in the autoimmunity laboratory. Clin Chem Lab Med 2006;44(10):1169-74.
7. Fritzler MJ. Advances and applications of multiplexed diagnostic technologies in autoimmune diseases. Lupus 2006;15:422-7.
8. Wiik A, Cervera R, Haass M, Kallenberg C, Khamashata M, Meroni PL, et al. European attempts to set guidelines for improving diagnostics of autoimmune rheumatic disorders. Lupus 2006;15:391-6.
9. Wiik AS. Anti-nuclear autoantibodies. Clinical utility for diagnosis, prognosis, monitoring, and planning strategy in systemic immunoinflammatory diseases. Scand J Rheumatol 2005;34:260-8.
10. Kavanaugh A, Tomar R, Reveille J, Solomon D, Homburger HA. Guidelines for clinical use of the nuclear antibody test and tests for specific autoantibodies to nuclear antigens. Arch Pathol Lab Med 2000;124:71-81.
11. Wiik AS, Gordon TP, Kavanaugh AF, Lahita RG, Reeves W, Van Venrooij WJ, et al. Cutting edge diagnostics in rheumatology: the role of patients, clinicians, and laboratory scientists in optimizing the use of autoimmune serology. Arthritis Rheum 2004;51:291-8.
12. Itoh Y, Rader MB, Reichlin M. Heterogenity of the Ro/SS-A antigen and autoanti-Ro/SS-A response: evidence of the four antigenetically distinct forms. Clin Exp Immuno 1990;81:45-51.
13. Bossuyt PM, Reitsma JB, Bruns DE, Gatsonis CA, Glasziou PP, Irwing LM, et al. The STARD statement for reporting studies of diagnostics accuracy: explanation and elaboration. Clin Chem 2003;49:7-18.
14. Bayer PM, Fabian B, Hubl W. Immunofluorescence assays (IFA) and enzyme-linked immunosorbent assays (ELISA) in autoimmune disease diagnostics -technique, benefits, limitations and applications. Scand J Clin Lab Invest Suppl 2001;235:(Suppl)68-76.
15. Dahle C, Skogh T, Aberg AK, Jalal A, Olcen P. Methods of choice for diagnostics antinuclear antibody (ANA) screening. Benefit of adding antigen-specific assays to immunofluorescence microscopy. J Autoimmun 2004;22:241-8.
16. Hoffman IEA, Peene I, Veys EM, De Keyser F. Detection of specific antinuclear reactivities in patients with negative anti-nuclear antibody immunofluorescence screening tests. Clin Chem 2002;48:2171-6.
17. Martins TB, Burlingame R, Von Mühlen CA, Jaskowski TD, Litwin CM, Hill HR. Evaluation of multiplexed fluorescent microsphere immunoassay for detection of autoantibodies to nuclear antigens. Clin and Dia Lab Immunology 2004;11:1054-9.
18. Shovman O, Gilburd B, Zandman-Goddard G, Yehiley A, Langevitz P, Shoenfeld Y. Multiplexed AtheNA multi-lyte immunoassay for ANA screening in autoimmune diseases. Autoimmunity 2005;38(1):105-9.
19. Rouquette AM, Desqruelles C, Larosche P. Evaluation of the new multiplexed immunoassays, FIDIS, for simultaneous quantitative determination of antinuclear antibodies and comparison with conventional methods. Am J Clin Pathol 2003;120:676-81.
20. Nifli AP, Notas G, Mamoulaki M, Niniraki M, Ampartzaki V, Theodoropoulos PA, et al. Comparison of a multiplex, bead-based fluorescent assay and immunofluorescence methods for the detection of ANA and ANCA autoantibodies in human serum. J Immunol Methods 2006;311:189-97.
21. Biagini RE, Parks CG, Smith JP, Sammons DL, Robertson SA. Analytical performance of the AtheNA MultiLyte ANA II assay in sera from lupus patients with multiple positive ANAs. Anal Bioanal Chem 2007;388;613-8.
22. Binder SR. Autoantibody detection using multiplex technologies. Lupus 2006;15:412-21.
23. Seideman J, Perrit D. A novel monoclonal antibody screening method using the Luminex-100 microsphere system. J Immunol Methods 2002;267:265-71.
_Salamunic_tablica4.jpg)



_Salamunic_tablica4.jpg)
