Introduction
An autoimmune disease occurs when the body's immune system begins to attack its own antigens. In general, these diseases are associated with humoral or cell-mediated immune reactions against one or more of the body’s own constituents (1). When self-tolerance is disturbed as a result of inflammatory pathogens, altered receptor, radiation or genetic background, it leads to a dysfunction of the immune system, which results in elevated autoantibody in the serum. The diagnosis of autoimmune diseases depends on the identification of disease-associated clinical symptoms and is associated with the detection of autoantibodies.
Clinicians conventionally classify autoimmune diseases as either systemic or organ-specific.
For systemic autoimmune diseases, varieties of autoantibodies are highly specific for certain diseases, including anti-dsDNA, anti-Sm, anti-ribosomal P autoantibodies in systemic lupus erythematosus (SLE), anti-topoisomerase I (Scl-70) in scleroderma, autoantibodies against citrulline-modified proteins (anti-CCP) in rheumatoid arthritis, anti-SS-A/Ro, anti-SS-B/La in Sjögren‘s syndrome (SjS), anti-U1-RNP, anti-PM-Scl in mixed connective tissue disease (MCTD) or anti-Jo-1 in polymyositis or dermatomyositis.
Organ-specific diseases are associated with autoantibodies specific to the main affected organ, like thyroglobulin (TGA) and thyroid peroxidase enzyme (TPO) in thyroiditis, insulin and glutamic acid decarboxilase autoantibodies in type 1 diabetes mellitus and anti-mitochondrial autoantibody in primary billiary cirrhosis. The autoantibodies may represent a status of disease activity or predict a future pathogenic condition.
Autoimmunity laboratories analyze and measure an increasing number of autoantibodies employing a broad spectrum of techniques and methods (2).
The first antinuclear antibody (ANA) detection method was the "LE cell" preparation test in 1947 by Hargraves, which linked autoimmunity to the systemic lupus erythematosus diseases (3). The immunofluorescence technique for detection ANA, which denotes specific subtypes based on the nuclear or cytoplasm component, was designed in 1957 (4). In the years following, laboratory scientists and the diagnostics industry have developed a variety of techniques and different methods for the detection of antibodies (5).
The first enzyme immunoassay method was introduced in 1972, and since that time several different forms of enzyme immunoassays have been developed, which were simple, high-throughput analyses and could be automated and standardized (6,7).
Traditional assays based on hemagglutination reactions, immunodiffusion and to some degree, immunofluerescence are increasingly being replaced by less demanding tests based on immunoblotting techniques or enzyme immunoassay (EIA), widely used for detecting the presence or concentration of individual autoantibodies in biological fluids. The newly developed multiplex immunoassay enables the simultaneous determination of different autoantibodies, in which a large number of antigens are immobilized on a solid carrier in spatial(planar) arrays orspectral (bead-based) arrays(8).
As soon as a new diagnostic or technical invention approaches and proves valuable, clinical diagnostic companies develop the new technology and commercialize it. By contrast, the development of standards is slower and requires consensus from the clinical diagnostics companies and the medical societies (9). Standardization of techniques and methods is important because it allows the comparison of results from different studies and improves the clinical interpretation of the findings. Standardization is possible if standard and analytes are identical, but biological samples are heterogeneous and differ from the standard.
Standardization of immunoassays methods for autoantibody detection is difficult due to the fact that pure standard, reference methods and standard reference materials (SRMs) are not available. Professional organizations of experts which are involved in immunoassays standardization accepted the problem and started the development of serum-based standards in which the matrix is similar to that in clinical samples. The specificity of an immunoassay is dependent mainly on the epitope specificity of the antigen that is used for determination of the autoantibody. Standardization is time-consuming and expensive, so clinical diagnostics companies often use a well respected commercial assay as a reference method. Large differences in assay calibration lead to an inadequate interpretation of the test results. By taking into consideration the standardization problems in immunology, the specificity of autoantibodies and antigens, the heterogeneity of biological material and by knowing the advantages and deficiencies of old and new technologies in the detection of autoantibodies, the laboratory could face a dilemma about detecting all clinically significant autoantibodies. Analyses of different test systems obtained in laboratories or in different clinical studies are not always interchangeable, which impairs evidence-based medicine (10).
Standardization of autoantibody assay "old" or "new" is critical to their use in the clinic to predict diagnosis and treat a very diverse group of autoimmune disorders.
Identification of human autoantibodies – responsibility of laboratory
Autoimmunity laboratories use immunoassays as the basic technique for the determination of autoantibodies and not the antigens (2). Important antigens have been well described and they are applied in methods that are used to detect autoantibodies. Antigens can be purified from nuclei of rabbit thymus or human spleen or recombinant. Many of the important autoantigens have been well characterized, which is important for the distinctiveness of the methods by which means autoantibodies are detected (10,11).
The detection from autoantibodies to antigens SS-A/Ro, SS-B/La, Sm, RNP, Scl-70, PM-Scl and Jo-1 is clinically useful in systemic autoimmune diseases.
The SS-A/Ro antigen is a ribonucleoprotein with molecular weight of 60 and 52 kDa. The autoantibodies present in the patients serum may be directed against both of the protein components.
The SS-B/La antigen is a 47 kDa phosphoprotein associated with a variety of small RNAs within the cell.
The Sm and RNP antigens are a group of heterogeneous molecules consisting of proteins which associate with small RNAs. Sm autoantibodies react with the 28 kDa protein B/B', and the 14 kDa D protein. RNP autoantibodies react most strongly with a 70 kDa protein and in some patients with the 32 kDa or 20 kDa C proteins. Because of the complex nature of these antigens, the methods used to produce these antigens for commercial tests are very critical.
The Scl-70 antigen is a 70 kDa protein, contains 765 aminoacids and has a topoisomerase activity. The recombinant Scl-70 is sensitive and has a high enzyme activity (4,6,7).
The PM-Scl antigen is a complex of 11-16 proteins with molecular weights varying from 20 to 111 kDa (4).
The Jo-1 antigen is identical to histidil-tRNA synthetase and is present in the cytoplasm. A major epitope is in the amino-terminal portion of the protein molecule.
There are a number of other antigens which have been used in assays for the detection of autoantibodies associated with specific systemic or organ specific diseases. The clinical usefulness of the analysis’ results depends on the quality of the laboratory tests. An ideal diagnostics test has both, high sensitivity and specificity. It also identifies all patients with diseases and is not positive in those who do not have diseases (11,12).
The laboratory may have dilemmas about techniques that are relevant and reliable to detect all clinically significant autoantibodies. At the same time, the applied method must be a tool for high throughput, efficient, easy to use and inexpensive (13,14).
It is responsibility of the laboratory to evaluate each new technique and methods with test sera from local patients. Quality management in clinical immunology laboratories include procedures used to ensure the accuracy and reproducibility of tests results and specification for autoantibody tests should include data on test validity, precision, cut-off value and their meaning, and test sensitivity and specificity, from which predictive values and likelihood ratio (LR) ratio can be generated. (15). This process needs to be attended by close collaboration of diagnostic laboratories, experienced clinicians who strive for an accurate diagnosis and patients (16).
Techniques and methods used for the detection of antibodies
Screening tests for autoantibody detection are performed for a variety of systemic and organ specific autoimmune diseases. Requests for these tests have risen remarkably, mainly due to the increased understanding of the nature of autoantibodies. A simultaneous development of new methods and analytical systems in clinical immunology has involved a constantly increasing expenditure of economic resources for the assay of antibodies (13). Inadequate use of laboratory tests is one of the most frequent problems in autoimmunity, leading to incorrect diagnoses and inadequate treatment (2).
Immunofluorescence
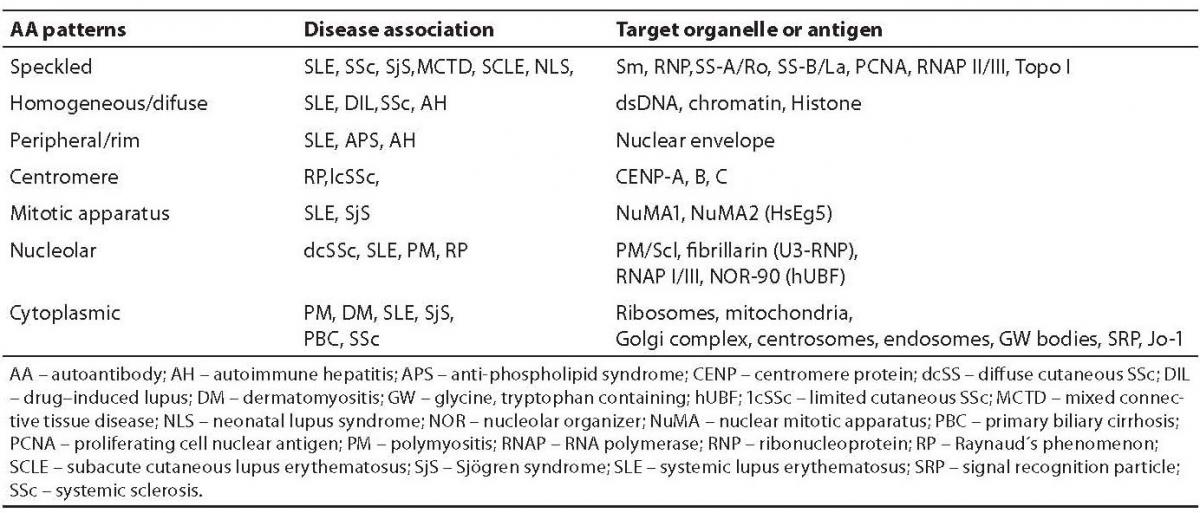
The indirect immunofluorescence (IIF) technique, which uses various tissue sections or the human tumor cell line (HEp-2) as an antigenic source, has had major implications for the diagnosis of autoimmune diseases in a routine laboratory setting (17). In IIF, undefined antigens are recognized by autoantibodies from the patient´s sera and yield specific patterns which must be interpreted in their relation to disease association (Table 1) (18).
Table 1. Autoantibody patterns and their relation to specific autoantibodies and disease
The IIF staining pattern of a positive sample can be used to evaluate which appropriate antigen specificities to look for. It is known that the HEp-2 cells used for the detection of autoantibodies do not have a satisfactory ability to give positive IIF results for antibodies to SS-A/Ro-52 and Jo-1 (histidil-tRNA synthetase) (19). Many serum samples give speckled or grainy homogenous staining patterns which cannot be clearly identified as one of the known patterns (17). The presence of various antigenic targets on the tissue section results in an excellent overall sensitivity. Antinuclear antibodies which have undefined specificities can be seen in the serum from patients with a wide variety of autoimmune diseases, infectious diseases and in some healthy individuals.
The lack of specificity may result in misleading interpretation and demonstrate the limitations of the IIF technique for screening purposes (6,11). Indirect immunofluorescence (IIF) microscopy is a sensitive method; yet, it has some limitations like substrate variations, manual performance, subjective result interpretation, low reproducibility and a lack of standardization. IIF is time-consuming, resulting in a low throughput and increased personnel costs. (20,30). In order to overcome this limitation, fully automated IIF interpretation systems with pattern-recognition software have been introduced recently. Standardization of autoantibodies testing by IIF remains a critical issue in and between routine laboratories and may be improved by automated interpretation systems (21).
The analysis of autoantibodies by IIF remains the hallmark of diagnosis, but some investigators have claimed that this technique is becoming out-of-date and that it could/should be replaced by enzyme immunoassays or multiplexed assays in the routine laboratory diagnostics (22).
Enzyme immunoassay
The enzyme-linked immunosorbent assay (ELISA) based either on antigens prepared from human tumor cell line (HEp-2) nuclear extracts and highly purified nuclear antigens or from recombinant antigens, has been the most promising, but the main differences, in terms of positively results, among various enzyme immunoassay methods have been described (23-26). The isolation of autoantigens from natural sources such as human tissues has major limitations with respect to reproducibility and purity. Many proteins are present only in limited amounts and their purification requires the removal of other potentially antigenic targets (17). The specificity of ELISAs for autoantibody measurements is strongly dependent on the quality of antigens used, and it is important that an antigen should have exactly the same sequence, conformation and post-translational modifications as the human antigen (20,27).
EIA is now widely used for identifying specific autoantibodies to nuclear or cytoplasmic antigens of different group of organ-specific disorders, such as Grave´s disease, primary biliary cirrhosis, insulin-dependent diabetes mellitus or systemic affecting different organs like systemic sclerosis, Sjögren’s syndrome, mixed connective tissue disease or rheumatoid arthritis (18,28).
The HEp-2 antinuclear antibody EIA (HEp-2 ANA EIA) is an automated method with high reproducibility and internal calibration as a basis for standardization. However, the evaluation of clinically well-defined samples in case of scleroderma patients with the use of HEp-2 ANA EIA for example yielded a lower rate of positive results compared to ANA IIF (26,29). Many studies conducted under standardized conditions showed the analytical variability of different test systems.
Multiplexed immunoassays
Multiplexed immunoassays support the identification of multiple autoantibodies from a single determination in the same time.
Microarray based assays
The line-blot immunoassay is a multiplexed immunoassay which allows the parallel analysis of different types of autoantibodies. Line assays use recombinant antigens almost exclusively which are immobilized in straight lines on a nylon test strip. When incubated with serum, autoantibodies that are present in the sample bound to the autoantigen lines on the strip. Bound autoantibodies are visualized with a color-detection system that relies on alkaline phosphatase activity. Results are interpreted by comparing the color intensities of the antigen lines with those of the cut off lines. Some publications have demonstrated that a few percent of IIF ANA negative sera will be positive using assay line assay, especially for anti-SS-A/Ro (10,31,32).
The planar microarray technology was developed and applied for the simultaneous detection of different autoantibodies using the sandwich immunoassay format. Different autoantigens are immobilized on a microarray along with control proteins. The arrays are subsequently incubated with patient sera and bound autoantibodies are detected by a labeled secondary antibody. Most of the microarray assays formats applied chemiluminescence or fluorescence based detection methods (33,34).
Bead-based assay
As an alternative to planar microarrays, flow cytometry for the analysis of bead-based immunoassays has been developed (31). Recently a commercially available microsphere-based fluorescent assay has been introduced for the detection of ANA. The potential low cost and time saving may be a reason for the routine use of these assays in the research and clinical laboratories (16). Immunoassay systems with micro-bead technology and flow cytometry detection (xMAP technology) have been applied to autoantibody measurement. The system uses polystyrene microspheres labeled internally with different ratios of two different fluorochromes. Each fluorochrome can have any of the 10 possible levels of fluorescence intensity, thereby creating a family of 100 spectrally addressed bead site. The antigens corresponding to autoantibodies are bound to the microspheres. Each of the 100 microbeads that can be differentiated by their fluorescence carries a specific immobilized antigen for a single autoantibody. At the same time, a green laser excites the external reporter fluorescence to quantify the specific reaction related to each autoantibody (35). Several companies supply commercial tests for simultaneous measurement of different autoantibodies by flow cytometry. The evaluation of a assay from different producers for the simultaneous quantitative determination in the same sample of nine antinuclear autoantibody specificities (dsDNA, SS-A/Ro, SS-B/La, Sm Sm/RNP, Scl-70, Jo-1, ribosome and centromere B) yielded good results (36). Clinical evaluation of a microsphere bead-based flow cytometry assays for simultaneous determinationof anti-thyroidperoxidase and anti-thyroglobulin antibodies showed good agreement with ELISA (31).
One problem in the detection of autoantibodies from patients is the lack of true quantitative calibration because of the different affinities of the antibodies to antigens (37). However, the major questions remain regarding whether the quantitative data obtained by multiplex bead-base assays are identical to, or at the least similar, to data obtained using other methods (35). Sensitivity, reliability and accuracy are similar to those observed with ELISA procedures (16,38).
Proteomic’s technologies for the study and diagnosis of autoimmune diseases
Clinical proteomics offers opportunities to identify new disease biomarkers in body fluids, cells and tissues. The focus of clinical proteomics in on the analytical and clinical validation and implementation of novel diagnostic or therapy related markers. (39)
Protein microarrays represent a validated platform for profiling protein levels and their post-translational modifications. Proteomic technologies including antigen microarray platforms enable the large-scale characterization of immune responses against forigen and self antigens that may be involved in the development and progression of autoimmune diease. Antigen microarrays allow the comprehensive analysis of autoantibodies directed against hundreds to thousands of antigens, including proteins, peptides, nucleic acids, macromolecular complexes (33).
Molecular probes can be monoclonal or polyclonal antibodies. Arrays are probed with cell culture, supernatant, cell lysate or serum. Depending on the molecule probe used, proteins or antibodies in the sample are bound to the planar array. The bound molecules are detected by a secondary antibody marked with fluorescent dye, or directly if the sample has been fluorescently labeled. The incubated chips can be read by a variety of scanners based on planar guide technology (34).
Although progress toward understanding the immune function has been made, the understanding of the underlying dysregulation and autoimmune response specificity remains limited. Alterations in genes that control pathways regulating self tolerance are critical in the pathogenesis of these diseases. The DNA microarray technologies are now available and providing a large number of information regarding the underlying pathophysiology of autoimmune diseases (40).
The application of proteomic techniques in diagnosing autoimmune diseases, predicting a disease course, treating with the proper therapy and monitoring the impact of the therapy will change the currently valid diagnostic procedure in the future (41).
Difference between methods
With the development of new technologies, there is a need to evaluate and standardize the technologies or diagnostic kits in an appropriate clinical laboratory setting (16).
Many studies conducted under standardized conditions showed the analytical variability of different test systems (29). Specificities and sensitivities of autoantibodies against different antigens are important for the diagnosis, but variability in results depend on the source of antigen, assays reproducibility, precision and accuracy and clinical manifestation of diseases. (10,42-45). Some studies showed agreement between IIF ANA and EIA (6,23-25,29), while others demonstrated differences in results (17,26,42,46). The choice of test is highly dependent on the clinical setting and higher sensitivity and specificity strongly depend on the cut-off value (6).
Multiplex technologies for the study of autoantibody profiles are the new technologies. The possibility of a simultaneous measurement of a number of correlated analytes (multiplexing) overcomes some limitations of conventional methods. However, the data do not correlate well with results obtained from IIF testing or EIA suggesting high rates of both false-positive and false-negative results (14,47-52). There are several substantial differences between multiplex bead-based assays and ELISAs. For example, the multiplex bead-based assay uses fluorescence as a reporter system where ELISAs use enzyme amplification of a colorimetric reaction. The multiplex bead-based assay captures ligands onto spherical beads in suspension while ELISAs generally rely upon flat surfaces in 96-well plates. The ELISA methods generally study one autoantibody at a time, while multiplex bead based assays are multiplexed and may be a subject to any perturbations that arise from analyzing multiple ligands simultaneously, such as cross-reactivities. (53).
There aremeasurable variations in the results obtained from assay kits obtained from different manufacturers (18,19). These methods are heterogeneous and different sources of antigens and different cut off values may contribute to a variability of results (5). More troubling is the observation that even if the same kits are used to detect antibodies of defined reactivity, there is an inter-laboratory variation of results (54).
The standardization of test results remains unsatisfactory
Results obtained in laboratories or in different clinical studies underline the need for a drastic standardization of the used procedures and the importance of independent calibrators or international standards.
Clinical scientists and assay manufacturers are challenged to provide quantitative and definitive autoantibody measurements based on reliable and reproducible assays which can provide clinically useful information with high specificity and sensitivity. The available diversity of test methodologies reflects the complexity of assays standardization (55).
Several organizations are involved in various aspects of immunoassay standardization. At the top of the standardization hierarchy is the International Standardization Organization (ISO). The World Health Organization (WHO) issues standards for clinically important antigens determined by immunoassay. In addition to these official bodies, several professional organizations have projects concerning immunoassay standardization, e. g. the International Federation of Clinical Chemistry (IFCC), International Union of Pure and Applied Chemistry (IUPAC), College of American Pathologists (CAP), National Committee for Clinical Laboratory Standards (NCCLS).
The Autoantibody Standardization Committee (ASC) was established in the early 1980s based on the recognized needs for reference human autoimmune sera in standardization (55). The European Autoimmunity Standardization Initiative (EASI) was formed to discuss how interaction between laboratories and clinical departments could be improved in practice, how algorithms in autoantibody testing could be harmonized and what an international concept of standardization of diagnostic strategies in this area should be considered (56).
Conclusion
Autoimmunity laboratories use immunoassays as the basic technique for the determination of autoantibodies. The central and main procedure principle for all autoantibody diagnostic assays is the capture of autoantibodies from serum using immobilized autoantigens. However, there is an enormous variability in these tests that has led to differences in results, a variable degree of confidence in their utility and even misdiagnosis of the patient´s disease (57). There is no universal solution to resolve these problems, but it is possible to improve the standardization level for techniques and methods. Standardization is an international problem and it is desirable that one international organization in collaboration with all organizations responsible for quality assessment of assays should be responsible for the coordination between diagnostic developers, clinicians and clinical diagnostics companies. In practice, the laboratories are responsible to solve the dilemma on suitable applications of respective methods for detecting autoantibodies in cooperation with the laboratory experts, clinicians and manufacturers.
Notes
Potential conflict of interest
None declared.
References
1. Lernmark A. Autoimmune diseases: are markers ready for prediction? J Clin Invest 2001;108:1091-6.
2. González-Buitrago JM, González C. Present and future of the autoimmunity laboratory. Clin Chim Acta 2006;365:50-7.
3. Hargraves M, Richmond H, Morton R. Presentation of two bone marrow components, the tact cell and the LE cell. Mayo Clin Proc 1948;27:25-8.
4. Cook L. New Methods for Detection of Anti-nuclear Antibodies. Clin Immunol Immunopathol 1998;88:211-20.
5. Rouquette AM, Desqruelles C, Larosche P. Evaluation of the new multiplexed immunoassays, FIDIS, for simultaneous quantitative determination of antinuclear antibodies and comparison with conventional methods. Am J Clin Pathol 2003;120:676-81.
6. Hayashi N, Kawamoto T, Mukai M, Morinobu A, Koshiba M, Kondo S, et al. Detection of Antinuclear Antibodies by Use of an Enzyme Immunoassay with Nuclear Hep-2 Cell Extract and Recombinant Antigens: Comparison with Immunofluorescence Assay in 307 Patients. Clin Chem 2001;47:1649-59.
7. Engvall E, Perlmann P. Enzyme-linked immunosorbent assay, ELISA. III. Quantitation of specific antibodies by enzyme-linked anti-immunoglobulin in antigen-coated tubes. J Immunol 1972;109:129-35.
8. Balboni I, Chan SM, Kattah M, Tenenbaum JD, Butte AJ, Utz PJ. Multiplexed Protein Array Platforms for Analysis of Autoimmune Diseases. Annu Rev Immunol 2006;24:391-418.
9. Bossuyt X, Louche C, Wiik A. Standardisation in clinical laboratory medicine: an ethical reflection. Ann Rheum Dis 2008;8:1061-3.
10. Eissfeller P, Sticherling M, Scholz D, Hennig K, Lüttich T, Motz M, et al. Comparison of Different Test Systems for Simultaneous Autoantibody Detection in Connective Tissue Diseases. Ann NY Acad Sci 2005;1050: 1-13.
11. Bossuyt X, Frans J, Hendrickx A, Godefridis G, Westhovens R, Mariën G. Detection of Anti-SSA Antibodies by Indirect Immunofluorescence. Clin Chem 2004;12:2361-9.
12. Von PAJM, Bast EJEG, Derksen RHWM. Cost-effective detection of non-antidouble-stranded DNA antinuclear antibody specificities in daily clinical practice. Reumatology 2006;45:629-35.
13. Tozzoli R, Bizzaro N, Tonutti E, Villalta D, Bassetti D, Manoni F, et al. Guidelines for the Laboratory Use of Autoantibody Tests in the Diagnosis and Monitoring of Autoimmune Rheumatic Diseases. Am J Clin Pathol 2002;117:314-24.
14. Salamunić I, Pauković-Sekulić B, Galetović A, Tandara L, Martinović-Kaliterna D. Comparative analysis of multiplex AtheNA Multi-Lyte ANA test system and conventional laboratory methods to detect autoantibodies. Biochem Med 2008;18:88-98.
15. Sturgess A, Edmonds J. Improving the effectiveness of autoantibody testing in the clinic. Autoimmun Rev 2002;1:273-8.
16. Fritzler MJ. Advances and applications of multiplexed diagnostic technologies in autoimmune diseases. Lupus 2006;15:422-7.
17. Hoffman IEA, Peene I, Veys EM, De Keyser F. Detection of Specific Antinuclear Reactivities in Patients with Negative Anti-nuclear Antibody Immunofluorescence Screening Tests. Clin Chem 2002;48:2171-6.
18. Stinton LM, Fritzler MJ. A clinical approach to autoantibody testing in systemic autoimmune rheumatic disorders. Autoimmun Rev 2007;7:77-84.
19. Wiik AS. Anti-nuclear autoantibodies. Clinical utility for diagnosis, prognosis, monitoring, and planing strategy in systemic immunoinflammatory diseases. Scand J Rheumatol 2005;34:260-8.
20. Haass M, Lehmann HP. New aspects of autoantibody detection with a new combination of autoantigenic targets. Clin Applied Immunol Rev 2001;1:193-200.
21. Hieman R, Büttner T, Krieger T, Roggenbuck D. Challenges of automated screening and differentiation of non-organ specific autoantibodies on HEp-2 cells. Autoimmun Rev 2009;9:17-22.
22. Dahle C, Skogh T, Åberg AK, Jalal A, Olcén P. Methods of choice for diagnostics antinuclear antibody (ANA) screening: Benefit of adding antigen-specific assays to immunofluorescence microscopy. J Autoimmun 2004;22:241-8.
23. Orton SM, Peace-Brewer A, Schmitz JL, Freeman K, Miller WC, Folds JD. Practical Evaluation of Methods for Detection and Specificity of Autoantibodies to Extractable Nuclear Antigens. Clin Diagn Lab Immunol 2004;11:297-301.
24. Fenger M, Wiik A, Høter-Madsen M, Lykkegaard JJ, Rozenfeld T, Hansen MS, et al. Detection of Antinuclear Antibodies by Solid-Phase Immunoassays and Immunofluorescence Analysis. Clin Chem 2004;50:2141-7.
25. González C. Guevara P, Alarcón I, Hernando M, Navajo JA, González-Buitrago JM. Antinuclear antibodies (ANA) screening by enzyme immunoassay with nuclear HEp-2 cell extract and recombinant antigens: analytical and clinical evaluation. Clin Biochem 2002;35:463-9.
26. Bizzaro N, Tozzoli R, Tonutti E, Piazza A, Manoni F, Ghirardello A, et al. Variability between methods to determine ANA, anti-dsDNA and anti-ENA autoantibodies: a Collaborative study with the biomedical indystry. J Immunol Methods 1998;219:99-107.
27. Binder SR. Autoantibody detection using multiplex technologies. Lupus 2006;15:412-21.
28. Jaskowski TD, Schroder C, Martins TB, Mouritsen CL, Litwin CM, Hill HR. Screening for antinuclear antibodies by enzyme immunoassay. Am J Clin Pathol 1996;105:468-73.
29. Tan EM, Smolen JS, McDougal JS, Butcher BT, Conn D, Dawkins R, et al. A critical evaluation of enzyme immunoassays for detection of antinuclear autoantibodies of defined specificities. Arthritis Rheum 1999;42:455-64.
30. Friou CJ. Clinical application of lupus serum nucleoprotein reaction using fluorescent antibody technique. J Clin Invest 1957;36:890-7.
31. Gonzalez C, Garcia-Berrocal B, Talavan T, Cassas ML, Navajo JA, Gonzalez-Buitargo JM. Clinical evaluation of a microsphere bead-based flow cytometry assay for the simultaneous determination of anti thyroid peroxidase and anti thyroglobulin antibodies. Clin Biochem 2005;38:966-72.
32. Gordon P, Khamashta MA, Rosenthal E, Simpson JM, Sharland G, Brucato A, et al. Anti-52 kDa Ro, Anti-60 kDa Ro, and Anti-La antibody profiles in neonathal lupus. J Rheumatol 2004;31:2480-7.
33. Joos TO, Stoll D, Templin MF. Miniaturised multiplexed immunoassays. Curr Opin Chem Biol 2001;6:76-80.
34. Wingren C, Borrebaeck CA. Antibody microarrays: current status and key technological advances. OMICS 2006;10:411-27.
35. Gonzales-Buitrago JM. Multiplexed testing in the autoimmunity laboratory. Clin Chem Lab Med 2006;44:1169-74.
36. Rouquete AM, Desgruelles C, Laroche P. Evaluation of the new multiplexed immunoassay, FIDIS, for simultaneous quantitative determination of antinuclear antibodies and comparison with conventional methods. Am J Clin Pathol 2003;120:676-81.
37. Feng Y, Ke X, Ma R, Chen Y, Hu G, Liu F. Parallel Detection of Autoantibodies with Microarray in Rheumatoid Diseases. Clin Chem 2004;50:416-22.
38. Seideman J, Peritt D. A novel monoclonal antibody screening method using the Luminex-100 microsphere system. J Immunol Methods 2002;267:165-71.
39. Apweiler R, Aslandis C, Defuel T, Gerstner A, Hansen J, Hochstrasser D, et al. Approaching clinical proteomics: current state and future fields of application in fluid proteomics. Clin Chem Lab Med 2009;47:724-44.
40. Robinson WH, Steinman L, Utz PJ. Proteomics technologies for the Study of Autoimmune Disease. Arthritis Rheum 2002;46:885-93.
41. Hueber W, Robinson WH. Proteomic biomarkers for autoimmune disease. Proteomics 2006;6:4100-5.
42. Tozzoli R, Bizzaro N, Tonutti E, Pradella M, Manoni F, Vilalta D. Immunoassay of Anti-Thyroid Autoantibodies: High Analytical Variability in Second Generation Methods. Clin Chem Lab Med 2002;40:568-73.
43. Lukinac Lj, Krilić D, Nöthig-Hus D, Kusić Z. The problem of thyroid antibodies testing. Acta Clin Croat 2004;43:355-9.
44. Vergani D, Alvarez F, Bianchi FB, Cancado ELR, Mackay IR, Manns MP, et al. Liver autoimmune serology: a consesus statement from the committee for autoimmune serology of the International Autoimmune Hepatitis Group. J Hepatol 2004;41:677-83.
45. Törn C, Mueller P, Schlosser M, Bonifacio E, Bingley P. Diabetes Antibody Standardization Program: evaluation of assays for autoantibodies to glutamic acid decarboxylase and islet antigen-2. Diabetologia 2008;51:846-52.
46. Rouquette AM, Desgruelles C. Detection of antibodies to dsDNA: an overview of laboratory assays. Lupus 2006;15:403-7.
47. Fritzler MJ, Behmanesh F, Fritzler ML. Analysis of human sera that are polyreactive in an addressable laser bead immunoassay. Clin Immunol 2006;120:349-56.
48. Biagini RE, Parks CG, Smith JP, Sammons DL, Robertson SA. Analytical performance of the AtheNA MultiLyte ANAII assay in sera from lupus patients with multiple positive ANAs. Anal Bioanal Chem 2007;388:613-18.
49. Shovman O, Gilburd B, Barzilai O, Shinar E, Larida B, Zandman-Goddard G, et al. Evaluation of the BioPlex 2200 ANA Screen Analysis of 510 Healthy Subjects: Incidence of Natural/predictive autoantibodies. Ann NY Acad Sci 2005;1050:380-8.
50. Avaniss-Aghajani E, Berzon S, Sarkissian A. Clinical Value of Multiplex Bead Based Immunoassay for Detection of Autoantibodies to Nuclear Antigens. Clin Vaccine Immunol 2007;14:505-9.
51. Shovman O, Gilburd B, Zandman-Goddard G, Yehiley A, Langevitz P, Shoenfeld Y. Multiplexed AtheNA Multi-lyte immunoassay for ANA screening in autoimmune diseases. Autoimmunity 2005;38:105-9.
52. Nifli AP, Notas G, Mamoulaki M, Niniraki M, Ampartzaki V, Theodoropoulos PA, et al. Comparison of a multiplex, bead-based fluorescent assay and immunofluorescence methods for the detection of ANA and ANCA autoantibodies in human serum. JIM 2006;311:189-97.
53. Elshal MF, McCoy P. Multiplex bead array assays: Performance evaluation and comparison of sensitivity to ELISA. Methods 2006;38:317-23.
54. Fritzler MJ, Wiik A, Fritzler ML, Barr SG. The use and abuse of commercial kits used to detect autoantibodies. Arthritis Res Ther 2003;5:192-201.
55. Chan EKL, Fritzler MJ, Wiik A, Andrade LEC, Reeves WH, Tincani A, Meroni PL. AutoAbSC. Org-Autoantibody Standardization Committee in 2006. Autoimmun Rev 2007;6:377-80.
56. Wiik A, Cervera R, Haass M, Kallenberg C, Khamashta M, Meroni PL, et al. European attempts to set guidelines for improving diagnostics of autoimmune rheumatic disorders. Lupus 2006;15:391-6.
57. Dodig S. Interferences in quantitative immunochemical methods. Biochem Med 2009;19:50-62.