Notification of critical values
Mario Plebani
[*]
[1]
Elisa Piva
[1]
Introduction
The last few decades have seen a significant decrease in the rates of analytical errors in clinical laboratories, and available evidence demonstrates that the pre- and post-analytical steps of the total testing process (TTP) are more error-prone than the analytical phase (1–3). In particular, most errors are identified in pre-pre-analytic and post-post-analytic steps outside the traditional laboratory environment, and beyond the direct control of the laboratory staff (4). Ordering and following-up onlaboratory and imaging tests consumes large amounts of physician time and is important in the diagnostic process. Now, diagnostic errors are the most frequent cause of malpractice claims in the US, and testing-related mistakes can lead to serious diagnostic errors (5). Recent data on errors in the pre-pre analytical phase underline that failures to order appropriate diagnostic tests, including laboratory tests, accounted for 55% of observed breakdowns in missed and delayed diagnosis in the ambulatory setting (6–8), and 58% of errors in the Emergency department (9). In the final steps of the loop, the incorrect interpretation of diagnostic or laboratory tests was found to be responsible for a high percentage of errors in the ambulatory setting as well as in Emergency departments (6–9). Improving quality in the post-analytical phase represents as such a valuable goal for reducing errors and improving patient safety. In particular, issues such as avoiding manual transcription of data, automated procedures for data validation and reporting as well as the implementation of systems which allows a more effective knowledge management to support data interpretation and clinical decision-making at the point of care may assure better quality and higher patient safety. A key-issue in post-analytical quality is represented by the effectiveness of laboratory data communication, particularly communication of critical test results (10)
Critical values: the concept
The concept of critical values was originally defined in 1972 (11) and elaborated on in 1990 (12) by George D. Lundberg, a pathologist who observed that very high or very low abnormal laboratory values indicated that the patient would die or have irreparable physical damage unless treated immediately. In the 30 years since Lundberg’s observations, critical values reporting received more focus and concern by the laboratory scientific community as well as by some regulatory organizations. The concept has been addressed in US legislation for clinical laboratories, well known as the Clinical Laboratory Improvement Amendments (CLIA), initially released in 1988. More recently, it was adopted by laboratory-accrediting agencies, such as the Joint Commission (JC) and the College of American Pathologists (CAP) in the US and by the Clinical Pathology Accreditation in the UK. The 2005 National Patient Safety Goals of the JC, in particular, include a requirement that health care organizations measure, assess and, if appropriate, take action to improve the timeliness of reporting, and the timeliness of receipt by the responsible licensed caregiver, of critical test results and values (13). More recently, the World Alliance for Patient Safety (WAPS) released 23 potential patient safety solution topics, and one of these, that is “communicating critical test results”, has been selected by the World Health Organization (WHO) International Steering Committee as a priority goal (14). Therefore, the definition and reporting of critical values is an important phase of the clinical laboratory testing process, and laboratories are responsible for detecting life-threatening results, for reporting them to health care providers, and also for tracking and improving the timeliness of reporting and the receipt of results. However, a consensus on several issues related to critical values and their communication does not exist. In particular, a great variability exists among the critical choice of the analytes, on the actual critical limits, on the professionals involved in critical values communication, both in reporting and receiving the data, and on the existence of written procedures and documentation.
Critical values as a controversial issue
Although it is widely agreed that clinicians must be informed of abnormal laboratory results that constitute a life-threatening condition for the patient as well as for any values for which delays in reporting can result in adverse outcomes for patients, the criteria for considering tests results critical are still largely controversial.
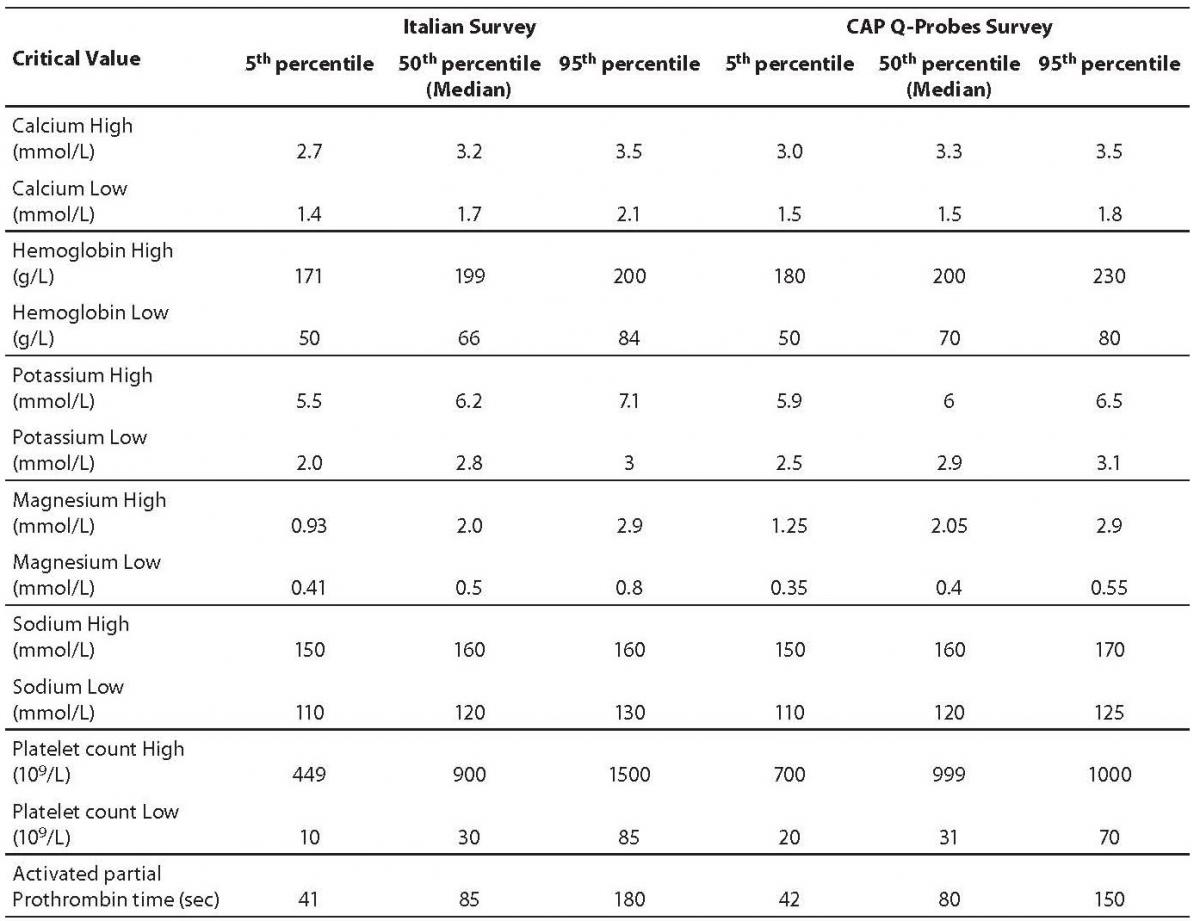
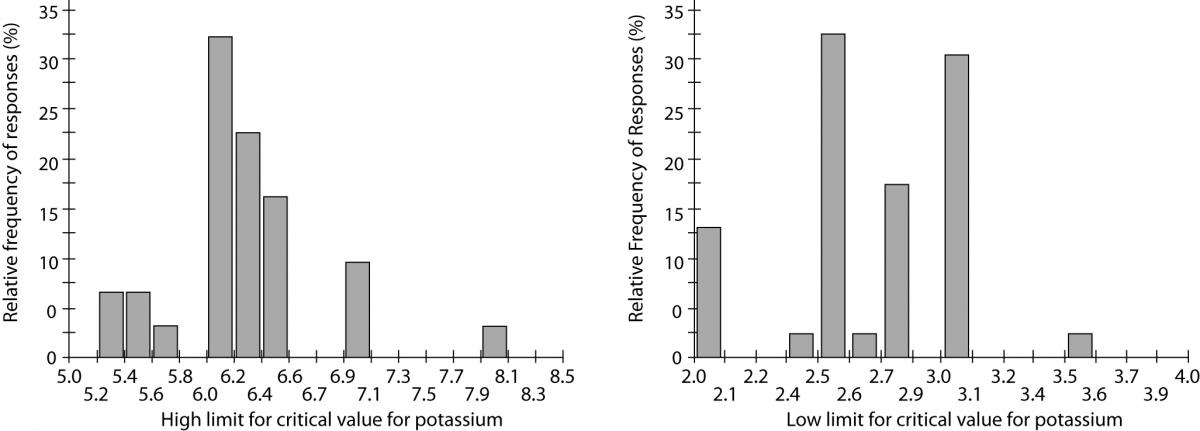
The first issue is the choice of the analytes. There is no consensus on the most reliable source of information regarding the list of critical values and clinical laboratories may follow both recommendations of scientific societies and clinician’s opinions in their institutions. The analytes more frequently included in the critical values list are in descending frequency: platelets, hemoglobin, potassium, sodium, calcium, prothrombin time (PT) or INR (International Normalized ratio), activated partial prothrombin time (aPTT), neutrophils, white blood cell count (WBC) and magnesium (15). Of these, sodium, magnesium, calcium and WBC were reported with low and high critical cut-offs by all participants. The INR and aPTT were reported with a single level (high cut-off) by all laboratories. Table 1 shows the distribution of cut-off values. Data are expressed as 5thpercentile corresponding to the lowest critical values, 50th that is the medianvalue, and 95th percentiles, among the Italian and US laboratories (Table 1, Figure 1).
Table 1. Critical value distribution. (Data for Italian Laboratories are from reference 15, for the US Laboratories from reference 16).
Figure 1. Inter-laboratory variability of critical value cut-offs for potassium. Relative frequencies for high (left side) and low (right side) limits. (Data from reference 15).
Reporting procedures
The reporting of critical values from the laboratory to caregivers is still made mainly by telephone (81.1% in the Italian survey). Less commonly used means of communication included computer (10%), fax (1.1%) or all tools indicated (15). In the US, laboratories have created call centers in order to centralize critical value reporting (17). In our institutions, we have introduced a computerized notification system using the laboratory (LIS) and hospital clinical information systems (HCIS). Briefly, once a critical value has been identified and validated by the clinical pathologist in charge, the transmission from the LIS to HCIS system creates an e-mail message for automated notification. On the HCIS patient record, this e-mail for critical value notification generates two actions: an SMS to the cell phone of the referring physician (both for in- and out-patients) and at the departmental level, an alert message by video to the ordering clinician. This automated communication has improved the timeliness of notification, and also prevented potential errors for which accreditation programs require read-back of the results. Moreover, it improved the likelihood of reaching the physician on call or the general practitioner and may provide important decision support (18).
Some interesting differences were observed when comparing the Italian and US policies in critical values notification. In Italy the notification is made principally by laboratory managers, i.e., pathologists, biologists, doctors on call, rather than by laboratory technologists, who provide the notification in only 11.1% of institutions (15). On the other hand, in the US institutions the professionals involved in communicating critical values are usually laboratory technicians or technologists who performed the tests (16).
In Italy, the critical values were communicated to physicians ordering the tests (37.3%), nurses (29.4%) and at a lower extent to any physician on call (17.9%), any people working in the ward (11.9%), and clerks (3%) (15). Again some differences with the US have been demonstrated, being the notification directed also to patients (12.2%), and even to the general practitioner’s relatives (16).
According to the inclusion of critical values communication among requirements of available standards for accreditation of clinical laboratories (19), 72.2% of Italian laboratories participating in the survey indicated that written procedures were in place in their institutions for critical values identification and reporting (15); 90% indicated that the timeliness of reporting is regularly assessed but only 63.35% of laboratories update their procedures at established times.
Conclusions
A recent study demonstrated that failures to inform patients on clinically significant abnormal test results or to document that they have been informed appear to be relatively common, occurring in 1 of every 14 tests (20). Failures involved common laboratory tests such as abnormal thyrotropin (T4), potassium, blood glucose levels and hematocrit. An even more recent study documented that 10.9% of critical patient care items were lost during a 24-hour observation period due to failures of communication (21). Among the categories of data lost in critical patient management, critical laboratory values and test results were most frequently lost during the control period (36.1% of lost observations). Improvements in communication of laboratory results, particularly critical results, seem to be needed for reducing errors and improving patient safety. Althoughthe concept of critical values has been introduced in the medical community many years ago, current data demonstrate a lack of harmonization both in defining the analytes as well as low and high critical values cut-offs. Moreover, the policies used for notifying critical values widely vary among laboratories in the same Country and when comparing the US with the Italian situation. It has been stressed that informatics may aid to reduce failure rates in notification of abnormal test results, namely crucial values (22). In addition, few data exist using laboratory critical values to evaluate variations in patient adverse events. In a recent study, however, critical values were plotted over time on statistical control charts and analyzed for unusual peaks in monthly occurrence rates, demonstrating the ability to rapidly detect adverse events, thus facilitating timely investigation (23). Therefore, efforts should be done for improving actual consensus on the definition and notification of laboratory critical values (e.g., that endorsed by the Italian Society of Clinical Biochemistry and Molecular Biology [SIBioC]) (24,25), and for evaluating their contribution to improve clinical outcomes and patient safety.
Notes
Potential conflict of interest
None declared.
References
1. Plebani M, Carraro P. Mistakes in a stat laboratory: types and frequency. Clin Chem 1997;43:1348-51.
2. Bonini P, Plebani M, Ceriotti F, Rubboli F. Errors in laboratory medicine. Clin Chem 2002;48:691-8.
3. Carraro P, Plebani M. Errors in a stat laboratory: types and frequenccies 10 years later. Clin Chem 2007;53:1338-42.
4. Plebani M. Errors in clinical laboratories or errors in laboratory medicine? Clin Chem Lab Med 2006;44:750-9.
5. Plebani M. Exploring the iceberg of errors in laboratory medicine. Clin Chim Acta 2009;404:16-23.
6. Hickner J, Graham DG, Elder NC, Brandt E, Emsermann CB, Dovey S, Phillips R. Testing process errors and their harms and consequences reported from family medicine practices: a study of the American Academy of Family Physicians National Research Network. Qual Saf Health Care 2008;17:194-200.
7. Gandhi TK, Kachalia A, Thomas EJ, Puopolo AL, Yoon C, Brennan TA, Studdert DM. Missed and delayed diagnoses in the ambulatory setting: a study of closed malpractice claims. Ann Intern Med 2006;145:488-96.
8. Wahls TL, Cram PM. The frequency of missed test results and associated treatment delays in a highly computerized health system. BMC Fam Pract 2007;8:32-42.
9. Kachalia A, Gandhi TK, Pupolo AL, Yoon C, Thomas EJ, Griffey R, et al. Missed and delayed diagnoses in the Emergency department: a study of closed malpractice claims from 4 liability insurers. Ann Emerg Med 2007;49:196-205.
10. Piva E, Plebani M. Interpretative reports and critical values. Clin Chim Acta 2009;404:52-8.
11. Lundberg GD. When to panic over abnormal values. MLO Med Lab Obs. 1972;4:47-54.
12. Lundberg GD. Critical (panic) value notification: an established laboratory practice policy (parameter). JAMA 1990; 263: 709.
13. Joint Commission on Accreditation of Healthcare Organizations. National Patient Safety Goals for 2006 and 2005. Available at: http//www.jointcommission.org/PatientSafety/NationalPatientSafetyGoals. Accessed January 8, 2009.
15. Piva E, Sciacovelli L, Laposata M, Plebani M. Assessment of critical values policies in Italian institutions: comparison with the US situation. Clin Chem Lab Med 2010; 48:461-8.
16. Wagar EA, Friedberg RC, Souers R, Stankovic AK. Critical values comparison: a College of American Pathologists Q-Probes survey of 163 clinical laboratories. Arch Pathol Lab Med 2007;131:1769-75.
17. Valenstein PN, Wagar EA, Stankovic AK, Walsh MK, Schneider F. Notification of critical results: a College of American Pathologists Q-Probes study of 121 institutions. Arch Pathol Lab Med 2008;132:1862-7.
18. Piva E, Sciacovelli L, Zaninotto M, Laposata M, Plebani M. Evaluation of effectiveness of a computerized notification system for reporting critical values. Am J Clin Pathol 2009; 131:432-41.
19. ISO 15189: 2007. Medical laboratories – Particular requirements for quality and competence.
20. Casalino LP, Dunham D, Chin MH, Bielang R, Kistner EO, Karrison TG, et al. Frequency of failure to inform patient of clinically significant outpatient test results. Arch Int Med 2009;169:1123-9.
21. Stahl K, Palileo A, Schulman CI, Wilson K, Augestein J, Kiffin C, McKenney M. Enhancing patient safety in the trauma/surgical intensive care unit. J Trauma 2009; 67:430-5.
22. Lippi G, Plebani M. Informatics aids to reduce failure rates in notification of abnormal outpatient test results. Arch Int Med 2009;169:1815.
23. Jenkins JJ, Mac Crawford J, Bissell MG. Studying critical values. Am J Clin Pathol 2007;128:604-9.
24. Lippi G, Giavarina D, Montagnana M, Luca Salvagno G, Cappelletti P, Plebani M, Guidi GC; SIBioC (Italian Society of Clinical Biochemistry and Clinical Molecular Biology); SIMEL (Italian Society of Laboratory Medicine); CISMEL (Italian Committee for Standardization of Laboratory and Hematological Methods); Inter-associative Study Group on the Extra-Analytical Variability of Laboratory Testing. National survey on critical values reporting in a cohort of Italian laboratories. Clin Chem Lab Med 2007;45:1411-3.
25. Simundic AM, Topic E. Quality indicators. Biochem Med 2008;18:311-9.