Introduction
Procalcitonin (PCT), the precursor of the hormone calcitonin, is a 116 aminoacid glycoprotein with a molecular weight of 14.5 kD (1). Under normal metabolic conditions, PCT is only present in the C cells of the thyroid gland, whereas in bacterial infections its concentration raises in blood. In healthy subjects, PCT concentration is < 0.1 μg/L and rises to levels above 100 μg/L during severe bacterial, parasitic or fungal infections with systemic manifestations, even in the absence of a thyroid gland. This indicates that PCT is mainly produced extrathyroidally in systemic inflammatory conditions (2). PCT determination was first performed in 1993 by Assicot et al. in children to differentiate bacterial from viral meningitis (3). Since that date, PCT has become a marker of bacterial infection with a wide range of indications for its use (4).
Systemic bacterial infection (sepsis) can be difficult to distinguish from localized infection and noninfectious conditions in critically ill patients, such as systemic inflammatory response syndrome (SIRS). Although infections and sepsis are accompanied by clinical and laboratory signs, such as changes in body temperature, leucocytosis, C-reactive protein (CRP) and tachycardia, they often provide low discrimination in the diagnosis of infection. In such cases early diagnosis of sepsis and a prompt antibacterial therapy are very important. Usually before the initiation of adequate therapy, blood culture analysis is performed in patients who are doubtful to have these conditions. Although blood cultures are very specific for the detection of sepsis, the overall sensitivity is only 25–42% (5). Furthermore, clinical signs of sepsis may develop without bacteriological evidence of infection and negative results do not exclude the presence of infection or sepsis (2). Thus, early diagnosis of sepsis and bacterial infection still depend on a sum of clinical criteria and microbiological procedures, which do not allow any conclusions on the extent, activity and prognosis of the disease.
As PCT is an established marker of severe systemic bacterial infection. The aim of this study was to evaluate the possible discriminative use of PCT in differentiating systemic and localized bacterial infection in critically ill patients. The main goals were to compare serum concentrations of PCT, together with other standard inflammatory parameters like CRP, platelet (PLT), white blood cell (WBC) and immature granulocyte (IG) counts, in patients with systemic and localized bacterial infection.
Material and methods
Patients
In our investigation we have included a group of patients hospitalized for > 1 week to the Intensive Care Unit (ICU) or to the Department of Burn Injuries of the University Hospital of Traumatology Zagreb, from July to November 2007 who fulfilled following inclusion criteria: clinical signs of sepsis (6) or localised infection. Patients were divided into two subgroups, according to the clinical signs and blood culture findings: patients with clinical sings of systemic bacterial infection and positive blood cultures comprised the group A (N = 11), while patients with localized bacterial infection and negative blood cultures but positive other cultures (urine culture, tracheal aspirate, upper respiratory tract swab), comprised the group B (N = 14). This study was approved by the Ethics Board of the University Hospital of Traumatology Zagreb.
Methods
Blood sampling for biochemical parameters and blood cultures was done on the day of onset of clinical sings of infection in all patients. In addition, in patients with negative blood cultures, samples for urine culture or tracheal aspirate or upper respiratory tract swab were taken according to clinical symptoms.
The biochemical laboratory analysis included the determination of PCT, CRP, PLT, WBC and IG count. Samples for PCT and CRP were centrifuged at 3000 x g for 10 minutes and serum aliquoted and stored at -20 °C until the analysis were done. PCT concentrations were determined in serum samples using enzyme-linked fluorescent immunoassay on the miniVidas analyzer (Biomerieux, Marcy I’Etoile, France), whereas CRP was measured in serum by immunoturbidimetric method on the Dimension Xpand (Siemens, Newark, DE, USA). PLT and WBC counts were determined in whole blood samples with K3EDTA as an anticoagulant on the automated hematology analyzer Sysmex XT-1800i (Sysmex Corporation, Kobe, Japan). Differential white cell count was performed manually in stained blood peripheral smears.
Statistical analysis
The Kolmogorov-Smirnov test was used to assess types of distributions of investigated parameters. Values were expressed as median and ranges. The Mann-Whitney U test was used for between-group differences testing. The Z-test was used for determination of the significance of difference between two proportions. The receiver operating characteristic (ROC) analysis was used to determine optimal cut-off values, area under ROC curve (AUC), with respective 95% confidence intervals (95% CI), specificity and sensitivity values. The values of P < 0.05 were considered statistically significant. Statistical analysis was done using MedCalc statistical software (MedCalc 10.3.0.0., Frank Schoonjans, Maraikerke, Belgium).
Results
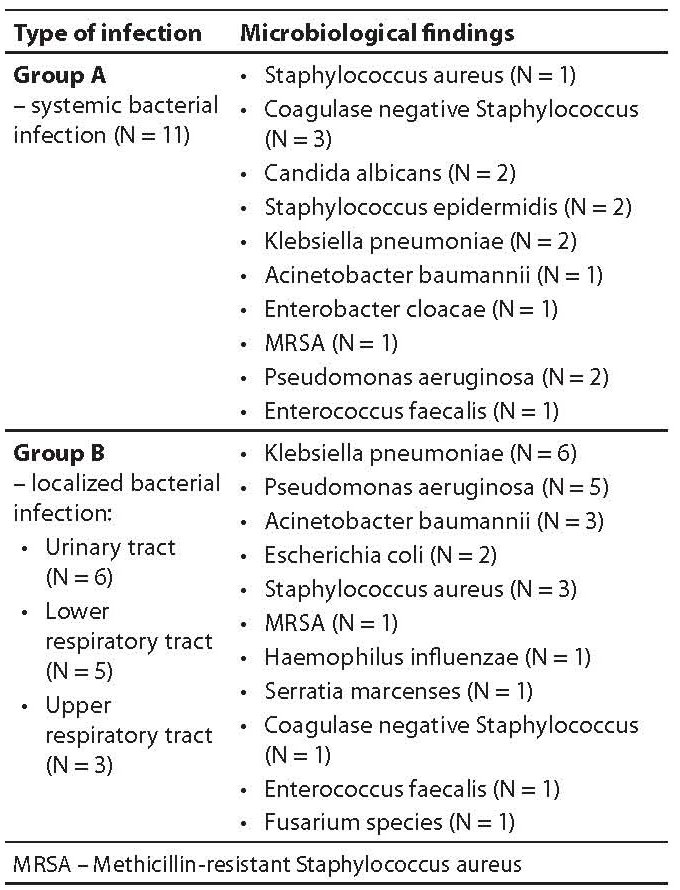
Type of infection and microbiological findings for studied groups of patients are presented in Table 1. Three patients in group A suffered from polymicrobial infection. Group B patients with localized infection according to microbiological and clinical results had urinary tract infection in 6 cases, lower respiratory tract infection in 5 cases (4 had polymicrobial infection) and upper respiratory tract infection in 3 cases (2 had polymicrobial infection).
Table 1. Type of infection and microbiology findings in groups A and B.
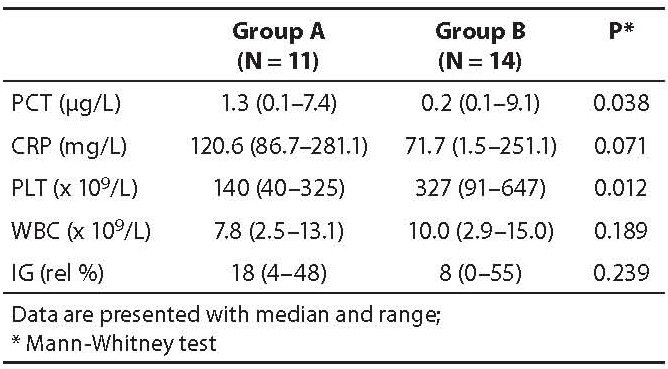
The obtained results of PCT, CRP, PLT, WBC and IG values are presented in Table 2. A statistically significant higher median value of PCT and a lower median PLT count was identified in group A as compared to group B (P = 0.038; P = 0.012, respectively). Although higher median values of CRP and IG and lower WBC were observed in group A than in group B, the differences between groups were not statistically significant (P = 0.071; 0.239 and 0.189, respectively).
Table 2. Measured parameters in all patient groups.
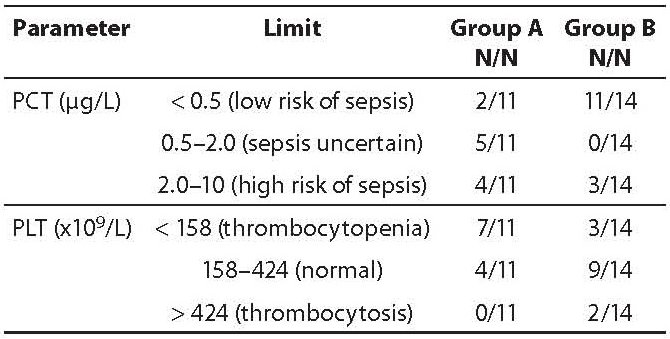
Table 3 presents patients in both study groups according to PCT limits recommended by the manufacturer and PLT values according to the reference interval (158–424 x 109/L). According to PCT limits, a low risk of sepsis was identified in the majority of group B patients (11/14) as compared to only 2/11 group A patients. Sepsis was uncertain in 5/11 group A patients, whereas 4/11 patients had a high risk of sepsis. In group B, none of the patients had uncertain sepsis but 3/14 of patients had a high risk of sepsis. In group A, 7/11 of patients had thrombocytopenia, while in group B a low PLT count was present only in 3/14 of patients. Although a higher number of patients with a combination of decreased PLT counts and elevated PCT concentrations was found in group A patients (6/11) as compared to group B patients (2/14), the difference between groups was not significant (P = 0.084) (data not shown).
Table 3. Statification of patients according to limits of PCT and PLT values.
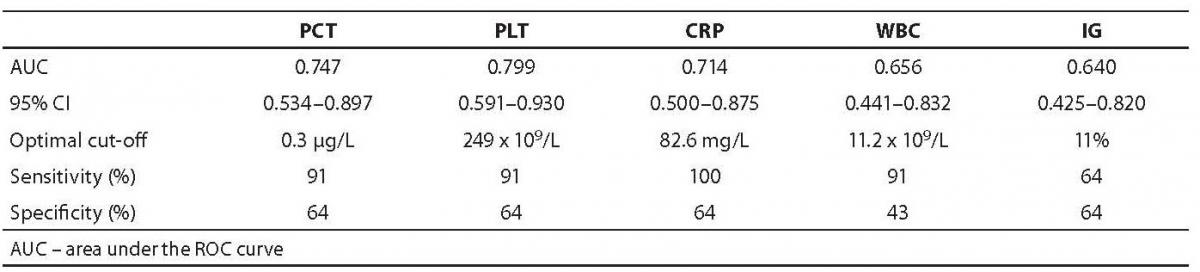
The results of ROC analysis (AUC with respective 95% CI as well as optimal cut-off values and their corresponding sensitivity and specificity) for all five measured parameters are presented in Table 4.
Table 4. Results of ROC analysis in the diagnosis of sepsis.
Discussion
Microbes like bacteria or fungi can cause severe diseases in humans. Bacterial infection may proceed from superficial colonization to localized invasive infection and result in further systemic manifestations such as sepsis. Since the disease may progress rapidly from a local process to systemic manifestations and organ dysfunction, time until diagnosis is very important in these patients (7,8). The main problems are that the diagnosis of sepsis in critically ill patients cannot be established using routine laboratory parameters and physical examination, while most confirmatory microbiological tests results are not immediately available.
Different studies and reviews have shown that PCT has a superior diagnostic accuracy for the diagnosis of sepsis as compared to other parameters and that a significantly increased PCT concentration in sepsis correlates with the severity of infection, response to treatment and prognosis (4,9,10). In our study, systemic infections were associated with a statistically significant PCT elevation as opposed to localized infections. Kofteridid et al. (11) found that PCT did not prove to be suitable for patients with less severe or localized infections, such as upper respiratory tract infections. In contrast, Becker et al. showed that PCT concentration may not always be related to systemic bacterial infectionandthat higher levels may occur in localized infections with negative blood cultures, such as in bacterial pneumonitis (12). Luzzani et al. (13) found that the median concentration of PCT in localized-infection patients was 1.3 μg/L (0.6–2.0) and in the septic group 3.1 μg/L (1.4–5.2). In contrast to Luzzani, in both groups of our patients PCT concentrations were lower. We can speculate that the possible reason for lower values in our patients with sepsis could be a previous history of sepsis in these patients, but we have no information regarding these data. According to Charles et al. (14), PCT concentration was significantly lower in patients with previous history of sepsis than in those without sepsis in their anamnesis. Another reason for lower PCT concentration in our analysis could be the fact that our study included only patients who spent at least a week in hospital after surgery or trauma in order to exclude PCT elevation due to non-bacterial causes in the first few days after trauma and surgical intervention (15,16).
According to the PCT limit for the presence of sepsis recommended by the manufacturer (< 0.5 μg/L), we have identified two cases with a low PCT but positive blood culture (Gram-positive bacteria). The explanation of this fact may be that infections with Gram-positive bacteria could produce delayed response as stated in study of Liaudat et al. (7). Another reasons could be previous antibiotic therapy or inadequate blood sampling time – prior to PCT elevation. In our study, when the infection was locoregional or confined to a single organ, the PCT concentration was low, except in three patients, who had polymicrobial infection of the lower and upper respiratory tract. High PCT concentration observed in these patients may indicate that PCT could be an important early marker for suspected septic patients, especially in case of negative blood-cultures. In such cases, PCT concentration should be helpful for better identification of subtle trends during the course of illness, but this has to be investigated in a larger number of patients. According to the ROC analysis (sensitivity 91%, specificity 64%) we obtained a lower cut-off (0.3 μg/L) than the value recommended by the manufacturer, which resulted in a higher number of false-positive results.
Data from the current literature have shown the role of CRP in the diagnosis of acute bacterial infection and for therapy monitoring, but most authors suggest usage of CRP in combination with other more acute biomarkers (13,17). Our data demonstrated that CRP induction occurred in almost all patients in group A and B with bacterial infection. In our patients optimal cut-off for differentiating sepsis from localized infection was 82.6 mg/L.
Sepsis is clearly one of risk factors for thrombocytopenia in critically ill patients and the severity of sepsis correlates with decrease in PLT count (18,19). In group A, 7/11 of patients had thrombocytopenia, and only 3/14 in group B. The median PLT count was significantly lower in patients with systemic bacterial infection. Decrease in PLT count can be caused by multiple and mostly interrelated factors. The mechanism by which thrombocytopenia occurs in sepsis includes PLT consumption and destruction, hemodilution, PLT sequestration (e.g., in the spleen) and their reduced production (20).
The WBC count and IG did not differ significantly across our patient groups. WBC and IG counts are simple and reliable measurements for detection and monitoring of inflammation, but they do not have a diagnostic benefit in ICU because both of them are unspecific (21). Most of the patients in group A and B had a normal or slightly higher WBC count with a left shift. Our data support the view of some authors that WBC count has little value in differentiating the type of infection in critically ill patients. Acute trauma, burns, seizures, and some medications can also have a significant effect on the WBC count (22).
We are aware of the limitations of our study, such as a small sample size and its limited potential impact to the obtained results. A second limitation is that our findings are based on a single measurement and sequential testing might improve the diagnostic value of one, or all of these parameters. Critics might argue that knowledge of the PCT value is unlikely to change clinical practice, because these patients are likely to receive antibiotic treatment based on clinical suspicion alone. However, we suggest that the value of this investigation lies not in confirming the suspicion of disease, but in helping to exclude systemic bacterial infection from the differential diagnosis. PCT detection should be included in the diagnosis strategy of critical ill patients. The advantage of PCT test is that results could be obtained very quickly (within 1 hour), unlike blood culture, the results of which are only available after 2 to 3 days.
In conclusion, our results obtained in small study groups have shown that PCT and PLT have better discrimination potentials than the other investigated standard inflammatory parameters for differentiating systemic from localized bacterial infection in critically ill patients. Further studies with larger patient samples and involving determination of inflammatory parameters in groups with systemic or localized infections should be carried out in order to confirm our results.
Acknowledgment
The authors would like to thank Mrs. Desiree Coen Herak for critical reading of manuscript and useful suggestions.
Notes
Potential conflict of interest
None declared.
References
1. Le Moullec JM, Julliene A, Chenais J, Lasmoles F, Guilana JM, Milhaud G, Moukhtar MS. The complete sequence of human preprocalcitonin. FEBS Lett 1984;167:93-7.
2. Reinhart K, Karzai W, Meisner M. Procalcitonin as a marker of the systemic inflammatory response to infection. Intensive Care Med 2000;26:1193-200.
3. Assicot M, Gendrel D, Carsin H, Raymond J, Guilbaud J, Bohuon C. High serum procalcitonin concentrations in patients with sepsis and infection. Lancet 1993;341: 515-8.
4. Christ-Crain M, Muller B. Procalcitonin in bacterial infections – hype, hope, more or less? Swiss Med Wkly 2005;135: 451-60.
5. Reimer LG, Wilson ML, Weinstein MP. Update on detection of bacteremia and fungemia. Clinical Microbiol Rev 1997; 10:444-65.
6. Bone RC, Balk RA, Cerra FB, Dellinger RP, Fein AM, Knaus WA, et al. American College of Chest Physicians/Society ofCritical Care Medicine Consensus Conference: definitions for sespsis and organ failure and guidelines for the use of innovative therapies in sepsis. Crit Care Med 1992;20:864-74.
7. Liadut S, Dayer E, Praz G, Bille J, Troillet N. Usefulness of Procalcitonin serum level for the diagnosis of bacteremia. Eur J Clin Microbiol and Infect Dis 2001; 20:524-7.
8. Kosanke R, Beier W, Lipecky R, Meisner M. Clinical benefits of procalcitonin. Tanaffos 2008;7:14-8.
9. Simon L, Gauvin F, Amre DK, Saint-Louis P, Lacroix J. Serum procalcitonin and C-reactive protein levels as marker of bacterial infection: A systematic review and meta-analysis. Clin Infect Dis 2004;39:206-17.
10. Meisner M, Adina H, Schmidt J. Correlation of procalcitonin and C-reactive protein to inflammation, complications, and outcome during the intensive care unit course of multiple-trauma patients. Crit Care 2006;10:R1.
11. Kofteridis D, Samonis G, Karatzanis AD, Fragiadakis GM, Bourolias CA, Maraki S, et al. C-Reactive protein and serum procalcitonin levels as markers of bacterial upper respiratory tract infections. Am J Infect Dis 2009;5:292-7.
12. Becker KL, Snider R, Nylen ES. Procalcitonin assay in systemic inflammation, infection, and sepsis: Clinical utility and limitations. Crit Care Med 2008; 36:941-52.
13. Luzzani A, Polati E, Dorizzi R, Rungatscher A, Pavan R, Merlini A. Comparison of procalcitonin and C-reactive protein as markers of sepsis. Crit Care Med 2003;31:1737-41.
14. Charles PE, Ladoire S, Snauwaert A, Prin S, Aho S, Pechinot A, et al. Impact of previous sepsis on the accurary of procalcitonin for the early diagnosis of blood stream infection in critically ill patients. BMC Infect Dis 2008;8:163.
15. Reinhart K, Karzai W, Hartog-Schier C. Procalcitonin (PCT) and its role in the diagnosis of sepsis. Sepsis 1998;2:157-61.
16. Whicher J, Bienvenu J, Monneret G. Procalcitonin as an acute phase marker. Ann Clin Biochem 2001;38:483-93.
17. Reny JL, Vuagnat A, Ract C, Benoit MO, Safar M, Fagon JY. Diagnosis and follow-up of infections in intensive care patients: value of C-reactive protein compared with other clinical and biological variables. Crit Care Med 2002;30:529-35.
18. Levi M. Platelet in sepsis. Hematology 2005;10:129-31.
19. Mavrommatis AC, Theodoridis T, Orfanidon A, Roussos C, Christopoulou-Kokkinou V, Zakynthinos S. Coagulation system and platelets are fully activated in uncomplicated sepsis. Crit Care Med 2000;28:451-7.
20. Vanderschueren S, De Weerdt A, Malbrain M, Vankersschaever D, Frans E, Wilmer A, Bobbaers H. Thrombocytopenia andprognosis in intensive care. Crit Care Med 2000;28: 1871-76.
21. Stevens DL, Root RK, Rosen H, Dale DC. Differential white blood cell count: underutilized or overrated. Curr Opin Infect Diseases 2000;13:205-7.
22. Hatherill M, Tibby SM, Sykes K, Turner C, Murdoch JA. Diagnostic markers of infection:comparison of procalcitonin with C reactive protein and leucocyte count. Arch Dis Child 1999;81:417-21.