The prevalence of metabolic syndrome in a local population in India
Manopriya Thiruvagounder
[*]
[1]
Shaheen Khan
[2]
Dhastagir Sultan Sheriff
[3]
Introduction
Metabolic syndrome (MS) also known as syndrome X is characterized by hypertriglyceridaemia and low concentration of high density lipoprotein (HDL) cholesterol (dyslipidemia), elevated blood pressure, impaired glucose tolerance and central obesity (1). People with MS are about twice as likely to develop cerebrovascular disease (CVD) and over four times as likely to develop type 2 diabetes compared to subjects without metabolic syndrome (2–4). It is well recognized that the syndrome has a genetic basis along with certain modifiable environmental factors (5,6). The present study was undertaken to report the prevalence of MS in the city of Salem, located in southern part of India.
Materials and methods
Subjects
Patients who attended the High Tech Hospital affiliated to Vinayaka Missions University, Salem, India for routine medical checkup formed the cases for the present cross-sectional study. The total of 1,568 cases that referred to the hospital during June 2007 to September 2009 was enrolled into the study. Informed consent was taken from all the subjects.
Metabolic syndrome was diagnosed according to the NCEP-ATP III criteria (Third Report of the National Cholesterol Education Program Expert Panel on Detection, Evaluation and Treatment of High Blood Cholesterol in Adults – Adult Treatment Panel III). Three or more of the following criteria formed the basis for defining metabolic syndrome:
1. hypertension, defined as a history of hypertension or a blood pressure greater than or equal to 130/85mm Hg;
2. hypertriglyceridaemia, defined as fasting triglycerides greater than or equal to 1.7 mmol/L;
3. hyperglycaemia, defined as a history of diabetes or a fasting blood glucose greater than or equal to 5.60 mmol/L;
4. low HDL-cholesterol, defined as a fasting HDL-cholesterol less than 1.00 mmol/L in men or less than 1.3 mmol/L in women; and
5. central obesity, defined as a waist circumference greater than 90 centimeters in Asian men or greater than 80 centimeters in Asian women (8,9).
Methods
The following parameters were collected: age, gender, waist circumference, blood pressure and fasting clinical chemistry parameters. Blood pressure was monitored and measured in a sitting position by a mercury sphygmomanometer (7). Blood samples collected after 12h overnight fast were analyzed to determine the concentrations of HDL-cholesterol, triglyceride and fasting glucose using Hitachi 901 Autoanalyzer (Roche Diagnostics GmbH, Germany) (8).
Statistical analysis
Data are presented as arithmetic mean ± standard deviation. Student’s t-test was used for comparison of numerical variables. Chi-square test was used to determine the level of significance for categorical variables. Multivariate logistic regression was used to define predictors of metabolic syndrome. Values of P < 0.05 were considered statistically significant.
Analysis was done using SPSS statistical software (Chicago, IL, USA).
Results
Out of 1,568 participants, there were 896 (56.8%) men and 672 (43.2%) women. Ten subjects did not participate in the study. The mean age was 49 ± 13 years (age range: 18–80 years), males: 50 ± 60 years; females: 53 ± 15 years.
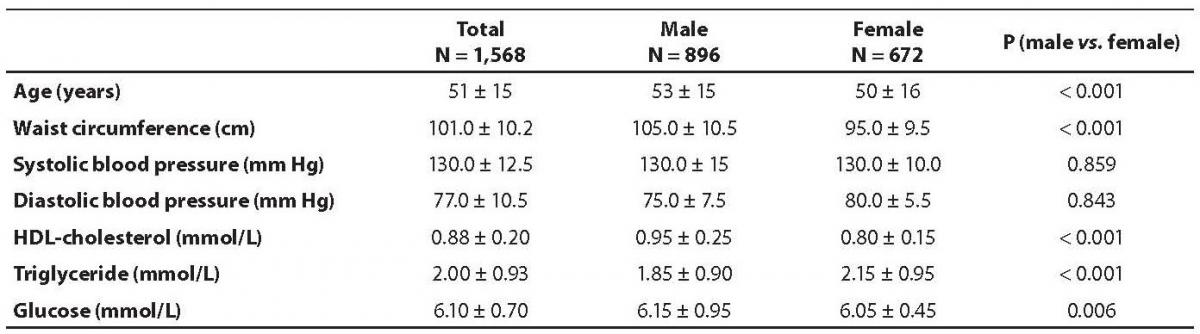
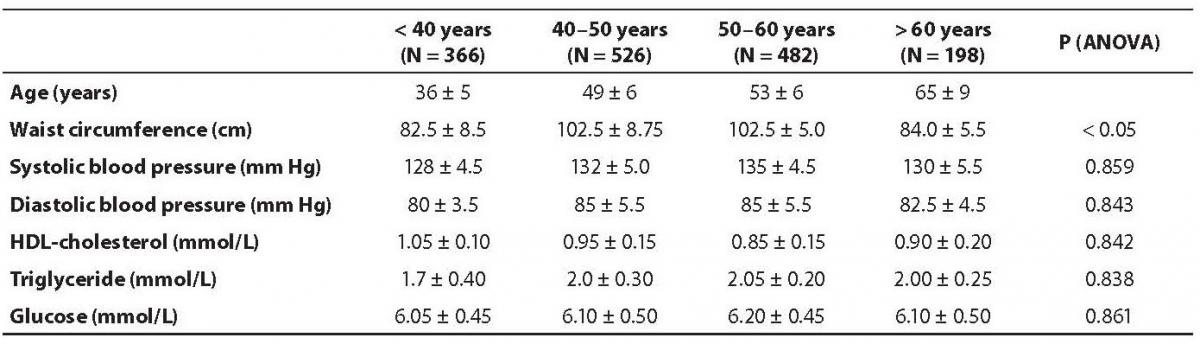
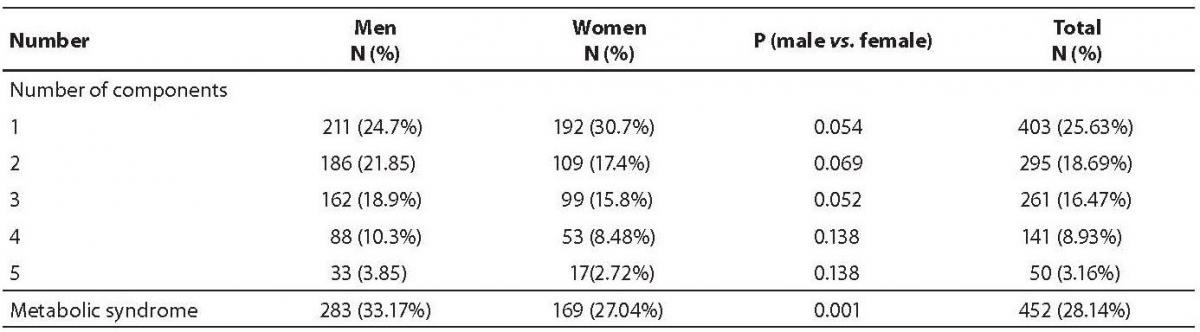
The demographic characteristics of the population are shown in Table 1. Abdominal obesity, dyslipidemia and impaired glucose tolerance were more prevalent in male subjects than in the females. None of the studied variables differed significantly between various age groups, with the exception of waist circumference (Table 2). The prevalence of metabolic syndrome was 33.17% in men and 27.04% in women (Table 3), the difference was statistically different (P = 0.001).
Multiple logistic regression analysis showed that increasing age (OR (95% CI) = 1.06 (1.040–1.070); P < 0.001) and male gender (OR (95% CI) = 1.50 (1.190–1.890); P < 0.05) were significant predictors of metabolic syndrome.
Table 1. Demographic characteristics of studeid population.
Table 2. Difference of clinical and biochemical parameters among age subgroups.
Table 3. Prevalence of components of metabolic syndrome.
Discussion
The National Cholesterol Education Program (NCEP) Adult Treatment Panel (ATP) III defined the criteria for the diagnosis of metabolic syndrome and laid down guidelines for its management (8). Insulin resistance is reported to be the key component for the clustering of risk factors that lead to metabolic syndrome (9).
In this cross-sectional population based study, 33% of men and 27% of women were diagnosed with the metabolic syndrome. Our results show that the prevalence of MS rises with age. Also, MS was more prevalent in men than in women. Asian Indians are known to have lower body mass index (BMI) than Europeans (10). However, for any given BMI, Asian Indians have greater waist-to-hip ratios and abdominal fat than Europeans (11,12).
It will be useful to measure factors associated with MS in different population to identify the prevalence of the syndrome and also to establish whether its prevalence differs according to region, religion and life style (13). It would be possible to prevent the early precipitation of diabetes or cardiovascular events in a community by finding out the presence of metabolic syndrome. This may help to establish basic guidelines of prevention and cure (14). It will be the best approach to formulate a health policy at a community level to provide health care and also educate the medical graduates to understand the basic health needs of the community they serve.
Notes
Potential conflict of interest
None declared.
References
1. Reaven GM. Role of insulin resistance in human disease (syndrome X). An expanded definition. Ann Rev Med 1993; 44:121-31.
2. BlatonVH, KoritaI, BuloA. How is metabolic syndrome related to dyslipidemia? Biochem Med 2008;18:14-24.
3. Meigs JB. Epidemiology of the metabolic syndrome Amer J Manag Care 2002;8:S283-92.
4. Bloomagarden ZT. American Association of clinical Endocrinologists (AACE) consensus conference on the insulin resistance syndrome. Diabetes Care 2003;26:1297-303.
5. Unger RH, Scherer PE. Gluttony, sloth and the metabolic syndrome: a roadmap to lipotoxicity. Trends Endocrinol Metab. 2010 Feb 23. [Epub ahead of print]. Doi:10.1016/j.tem.2010.01.009.
6. Bogardus C, Lillioja S, Mott DM, Hollenbeck C, Reaven GM. Relationship between degree of obesity and in vivo insulin action in man. Amer J Physiol 1985;11:E286-91.
7. Expert Panel on Detection, Evaluation of and Treatment of High Blood Cholesterol in Adults. Executive summary of the Third Report of the National Cholesterol Education Program(NCEP).Expert Panel on Detection, Evaluation and Treatment of High Blood Cholesterol in Adults(Adult Treatment Panel III) JAMA 2001;285:2486-97.
8. Inoue S, Zimmet P. The Asia-Pacific Perspective: Redefining obesity, and its treatment. Health Communications Australia 2000:17-20.
9. Third Report of the National Cholesterol Education Program (NCEP) Expert Panel on Detection, Evaluation and Treatment of High Blood Cholesterol in Adults (Adult Treatment Panel III) final report. Circulation 2002;106:3143-421.
10. WHO Expert Consultation. Appropriate body-mass index for Asian populations and its implications for policy and intervention strategies. Lancet 2004;363:157-63.
11. Raji A, Seely EW, Arky RA, Siminson DC. Body fat distribution and insulin resistance in healthy Asian Indians and Caucasians. J Clin Endocrinol Metab 2001;86:5366-71.
12. Park YW, Zhu S, Palaniappan L,Heshik S, Carnethon MR, Heymsfield SB. The metabolic syndrome prevalence and associated risk factor findings in the US population from third National Health and Nutrition Examination survey, 1988– 1994. Arch Intern Med 2003;163:427-36.
13. Bray GA, Jablonski,KA, Fujimoto WY, Barrett-Connor E, Haffner S, Hanson RL, et al. Relation of central adiposity and body mass index to the development of diabetes in the Diabetes Prevention Program. Amer Jour Clin Nutr 2008;87: 1212-8.
14. Knowler WC, Barrett-Connor E, Fowler SE, Hamman RF, Lachin JM, Walker EA, et al. Reduction in the incidence of type 2 diabetes with lifestyle intervention or metformin. New Eng J Med 2002;346:393-403.