Introduction
Overcrowding and prolonged length of stay (LOS) in emergency departments (ED) are an increasing problem in public hospitals (1). EDs and their payers (Governments or private providers) are under pressure to reduce LOS, improve health outcomes, and reduce costs. Although the operations of emergency services are complex, the availability of ancillary services, including clinically appropriate turnaround time (TAT) for laboratory tests, influences patient care decisions. For example, a 60 minute collection to validation TAT for general chemistry (electrolytes, liver function tests, and troponin) is the generally agreed acceptable TAT by laboratories for EDs and other critical care units (2,3).
Although there are many variables contributing to the increasing number of admissions to EDs, presentations from drug overdoses; either intentional or accidental (prescription, non-prescription or illicit substances) are increasing due to the wide availability of drugs of abuse (DOA) and the frequently changing armamentarium of natural, synthetic and recreational drugs. To aid clinicians in diagnosis and management, urinary drug screens (UDS) are widely used. Ideally, DOA results should be available at the same time as chemistry results for best patient care decisions. This is because rapid availability of all these results has an impact on multiple outcomes including clinical decision-making, admissions or discharge decisions and resource utilisation.
Generally UDS are performed in cases where the patient has or is reported to have a history of ingestion, presents with specific symptoms or has experienced trauma (4). In our organization, we became aware of an increasing number of gas chromatography/mass spectrometry (GCMS) drug screens being undertaken. Additionally, in some instances, our GCMS turnaround time had blown out up to 33 days, many weeks after the discharge of the patient. It was thus unclear how much benefit our drug screening process was improving patient care. We hypothesized that there was over-servicing of urine tests for drugs of abuse testing, causing the organization unnecessary cost without clear and timely clinical benefit.
The aim was to assess the need for routine GCMS in screening for drugs of abuse in a tertiary referral centre.
Materials and methods
Medical facilities, patient types and testing processes in place
Our organisation (Pathology Queensland) operates 33 pathology laboratories with nine of these serving as major hospital service providers. Samples for DOA screening are received via EDs, inpatient medical units, psychiatric units and various drug dependency/rehabilitation clinics. The nine major laboratories provide an immunoassay DOA panel on the general chemistry analysers (Beckman Coulter Brea, CA, USA). This consists of amphetamine like substances, benzodiazepines, cannabis metabolites, cocaine metabolites, methadone and opiates. Additionally, a confirmatory chromatography screening service to examine an extended array of drugs is offered (GCMS). The GCMS services provided are only qualitative which is used to confirm DOAs IA positives (and negatives) by identification of specific drugs and their metabolites, as well as to provide a toxicology service for general drug enquiries. Some of the limitations are that services cannot routinely detect and identify large molecular weight, highly polar and thermally labile drugs. The range of drugs able to be detected can be extended by chemical derivatisation, however this is not routinely performed. Overall, it detects many hundreds of prescription and over-the-counter drugs as well as certain poisons (e.g. hyoscine and hyoscamine from datura, strychnine, caffeine and nicotine in infants, for example). Clinicians could order either panel (rather than an initial screen) and a confirmatory GCMS test, regardless of the results of the IA test.
An audit undertaken pre-change (unpublished data) showed that 39.1% of drug requests for IA were also receiving a confirmatory GCMS test, regardless of whether it had been requested. This audit was undertaken via a standard “extended” search of our laboratory information system to extract the patient samples undergoing IA DOA and GCMS testing. Where both were undertaken, the pathology request form was reviewed to check if GCMS had been requested. The cause of the additional requesting in the laboratory was not analysed.
Service impact
The ease with which a full GCMS screen was available was overwhelming the chromatography unit performing the analysis, affecting TAT in some cases taking up to 33 days. The increasing workload, laboratory resource consumption and clinician demand for better TAT of results began to have a significant effect on the morale in our laboratory staff. When the audit results became available, a consultative process was undertaken to determine why GCMS was requested so frequently as a frontline panel, and what could be done to implement a change and improve service.
Consultative process
The process commenced by consultation with clinicians and clinical units most frequently ordering GCMS. The questions in the consultation process were:
· Why chromatography?
· Has an IA been performed?
· If IA completed, what additional information was hoped to be gained from chromatography?
· When was the data required?
· Will the results change medical management?
It was noted that almost all of the twenty clinicians (junior medical staff) interviewed had no understanding of the DOA screening and the difference in the information generated between GCMS and IA. At one large urban Drug and Alcohol Clinic it appeared that none of the medical attendees had a working knowledge of the laboratory tests being requested, but this was not formally assessed.
A decision was made to issue a directive Pathology Queensland wide that all requests for drugs of abuse testing would have a standard IA test undertaken, unless there was a specific request on the pathology form for GCMS, or the samples were from neonates or for juvenile drug testing. Samples would not be routinely tested with GCMS; however, the samples were to be held for 7 days so that a clinician could request a GCMS should it be required retrospectively.
Specimens for medico-legal or forensic needs were required to be packaged in line with chain-of-custody guidelines and forwarded to a forensic toxicology laboratory. Additionally, guidelines for DOA and toxicology investigations were developed and appended with all results to inform clinicians of the change and the new procedures. Furthermore, these most frequently requesting clinicians and units were directly verbally informed and then informed in writing of the change. This allowed clinicians to determine and specify on the request form if they had a need to look for additional and specific drugs e.g. newer “street” drugs, patient compliance for a single drug, for example.
Testing effectiveness
For the chromatography unit to improve TAT and for results to have greater clinical relevance when patients are in most need (overdose), it had to improve TAT. This was something that could not be easily accommodated, without a new policy. Further, there were concerns regarding any clinical or legal sequelae (e.g. for patients taking drugs of abuse whilst on an opioid maintenance programme) if an IA drug screen was undertaken as the initial test, and GCMS was not routinely performed. Since GCMS could still be undertaken as initial test if the requesting doctor informed the laboratory of the relevance of that test, effectiveness was measured as in two ways - one was the number of either complaints or calls to the Pathologist hotline querying why an IA and not a GCMS test was undertaken; and the second was phone calls to the 20 most common ‘users’ of GCMS as an initial tool asking for clinical outcomes and feedback on the new policy.
Results
Consultative process
A total of 819 GCMS samples were audited and the 10 most frequently requesting doctors and clinical units were consulted. These were mostly drug dependency clinics and some ED and Mental Health units. Their main reason for requesting GCMS was that immunoassays (IAs) were not as sensitive, especially at low concentrations (which although not having a clinical effect on the patient could indicate diversion with medico-legal implications). There were other reasons less commonly cited including cross-reactivity, medical reasons e.g. patient stopping therapies, the need for knowledge of presence of buprenorphine or medico-legal tests. Pathology Queensland agreed that where there was one of these indications, GCMS would be undertaken.
Numbers of requests and pattern analysis
The five yearly sample numbers pre and post-test ordering protocol change for IA and GCMS are shown in Table 1.
Table 1. Number of samples over time period: pre and post-test ordering protocol change. Change in ordering practice was in the mid March, Year Three.
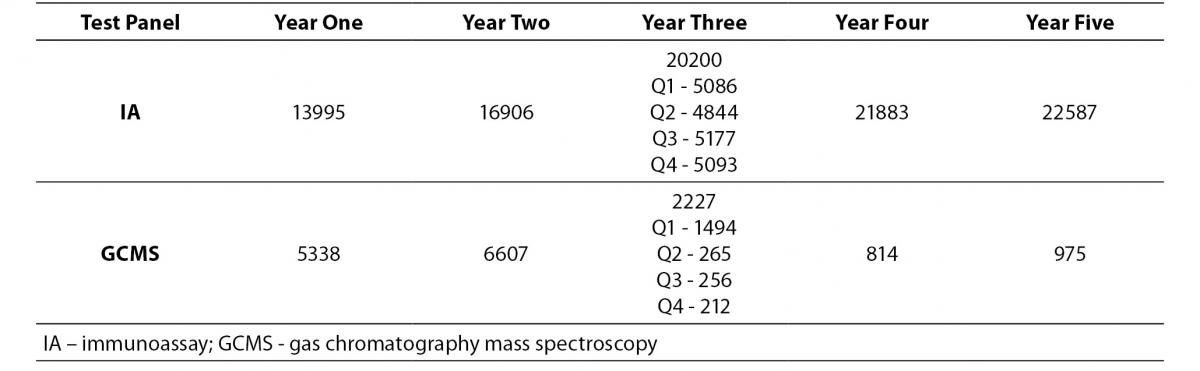
A subset of samples in each 3-month period of IA and GCMS testing results were examined before the change and after the change. Further, GCMS requests were compared with IA results to examine false negative and false positive results. Pathology request forms were examined for clinical indication. It should be noted that there was a much higher proportion of IA drugs confirmed on GCMS in the post change set for amphetamines (pre 70.1%, post 76.4%) and methadone (pre 92.7%, post 97.5%) and lower proportion for benzodiazepines (pre 89.6%, post 78.9%), and opiates (pre 94.4%, post 90.4%) compared to the pre change set. The data suggested there was minimal difference in detecting the presence of the selected drugs between IA as GCMS methodologies even though GCMS analysis post positive IA would only be undertaken if there was a requested clinical indication. Audit of the post change set requests showed only 4.3% of drug requests for IA were also receiving confirmatory GCMS testing, compared to 39% prior to intervention.
Drugs that were not detected by IA but confirmed by GCMS were likely to be due to drugs present below the set cut-off limits for the IA, as specified by Pathology Queensland and based on the AS/NZS 4308:2008 standard (Australian and New Zealand Standard providing cut off limits for drugs). The drugs detected by IA but not confirmed by GCMS are likely to be due to cross-reactivity of the immunoassay methods, resulting in false positive results (5). Although some of the drugs not confirmed by GCMS have been tested up to defined concentrations by Beckman (Beckman Coulter, Brea, CA USA), they (or their metabolites) may cross-react above these concentrations sufficiently to produce a detectable value.
The mean TAT (registered to validated) of results from two of our hospitals was also audited. From our second largest hospital, Princess Alexandra Hospital (PAH), and our most distant hospital (~1800 km), Cairns Base Hospital (CBH) which services distant and dispersed remote communities, and small towns that may take days to send the samples to the laboratory. The audit data for PAH samples showed the TAT was 3-4 days for GCMS and < 70 minutes for IA results. The audit data for CBH which includes samples from the remote sites was 3-4 days for GCMS and ~13 hours for IA results, but samples originating from the CBH hospital had similar TATs, < 70 minutes.
Clinical outcome
Whilst it was suggested that not routinely undertaking GCMS could lead to a reduction in patient care, analysis of the results show this was not borne out, bearing in mind the limitations of the clinician review of this retrospectively collected data for this study. Post review change showed less than 1% (0.63% amphetamines, 0.8% benzodiazepines) with a negative IA had a positive GCMS for a drug of abuse. These are likely to be due to concentration too low to be detected on IA. 1.9% (methadone – predominantly methadone detected on GCMS), and 10% opiates (predominantly buprenorphine) were not detected on IA yet detected on GCMS. It is known that buprenorphine is not detected on the particular IA used within Pathology Queensland and clinicians have been advised to request a GCMS if this drug is suspected. However, of these patients, none would have had a change in their acute care prior to the GCMS being undertaken. Buprenorphine testing does not generally come into play with acute or ED patients. It is more applicable to ATODS clinics. Our results suggested the IA methods provided the required information on the presence or absence of the drugs of most interest in almost all cases when compared with GCMS, allowing clinicians to utilize the data in unison with other pathology results and clinical information in real time to best manage patients. The commonest feedback from clinicians was increased confidence in the IA results, which allowed speedier decisions on clinical management; improved time management – specifically time required by medical staff in attempts to extract information on drug consumption from unwilling and incapable patients and relatives in the consultative phase, leading to improvement in productivity. The availability of the results in improved TAT thus provides confidence in decision making, more in line with evidence based practices.
Cost savings
In the two years prior to the change, ~39% of IA samples had a GCMS screen. In contrast post change ~4% of samples had a GCMS screen. The cost to the clinical units of the 44,471 sample numbers requested for IA DOA in year 4 and 5 was ~$1.62 M (AU$) while the cost of the 1790 GCMS sample numbers was ~$80,000. If the pre change practice was continued, an additional 35% (15,565 samples) analysed by IA DOA would have had GCMS performed, costing additional ~$127,000 over the two years.
Furthermore to have continued with the existing service and improve it, an additional staff member (~$100,000/year) and an additional GCMS system (~$1400,000) would have been required.
Discussion
In this study we have shown that changes in behaviour around test requests were possible without obvious clinical harm. Despite the limitations of the methodology to retrospectively adjudicate clinical decision making, there were other clinical benefits reported due to the provision of improved TATs - necessary to make timely clinical decisions. A TAT over a month is a clinically irrelevant result, unless the test is required for medico legal reasons or concerns regarding specific drugs that would be unlikely to be detected with an IA (for which it was still possible to request a GCMS). The other gain was a large financial saving for our organization, which could then be directed towards other needs. Specifically, the practice change has freed up two scientists to undertake other clinical work.
This laboratory-led change has been sustained – with changes persisting up to three years later. It is interesting that apart from phone advice when clinicians rang pathology; there was no ongoing formal education process around test requesting or interpretation.
This study is important for two reasons. Firstly, it shows that behavioural change and improved pathology turnaround time is possible. Secondly, it demonstrates how improved efficiency of laboratory processes can improve costs significantly, both directly and indirectly viaimproved patient TAT. In other studies it has been shown that laboratory services may make up 5% of a hospital’s budget but facilitate 60-70% of all critical decision-making such as admittance, discharge, and medication (6). More recent publications estimate this to be as high as 80-90% (7). Approximately 80% of complaints laboratories receive are about TAT, even though there are differences amongst clinicians on what constitutes acceptable TAT (8). It is estimated that 87% of dissatisfaction complaints originate from EDs, the place where most DOA testing is undertaken (9). These key points make laboratories pivotal in introducing change in test ordering patterns and behaviour. Laboratories know the intricate details of their costs (labour, equipment, reagents, performance, etc.), TATs, test details and have a high level of knowledge on the relevance of tests for patient care. Laboratories can guide the multidisciplinary team on efficient and effective utilization of their services. However, laboratories are often reluctant to impose decision on clinical experts. This work has shown that it is possible to lead a change from Pathology and that is sustainable on long-term basis. It is becoming more and more important that healthcare teams look for novel ways to provide timely, cost effective, and clinically applicable services so that clinicians are able to make evidence based patient care decisions. Thus, interventions like in this study are necessary and pathology services will need to be proactive drivers.
Drug-testing is perceived to have a low impact on therapy if not provided in good TAT. In fact, most poisoned patients are managed symptomatically and supportively regardless of the drug or poison’s identity (10) with timely urine DOA testing useful if antidote or specific therapeutic intervention is available, such as naloxone for opiates (4). Fabbri et al. similarly concluded that the use of multidrug analysis is questionable in the management of patients with acute drug poisoning, although it is rather useful to reassure the clinician of their actions (11). Other publications advocate such tests are not necessary as the clinical course of patients with suspected drug overdose can be predicted during the first few hours of observation (12). In some patients, such as those with altered mental status, where the medical history may be unclear at the time of presentation to the ED, drugs of abuse tests do aid clinicians in diagnosis and management of drug-related complications (13).
Other pathology services have also tried to manage improvement in the TAT by introducing point-of-care drugs of abuse testing. A recent example of this has shown a $132,951 ($US) increase in cost but a savings of 15.5 hours per day bed time (14). However, this result could be considered as cost shifting savings only (i.e. from laboratory to the clinical unit) unlike our study which is pure cost savings. Equally it needs to be pointed out that the laboratory is far better equipped at ensuring highest quality assurance is achieved with results. Performing urine drug screens by IA methods on existing general biochemistry analysers provides a very rapid and cost effective mechanism. However, as seen in this study, IA methods can produce false positive or negative results. Hence, the availability of confirmatory drug testing by chromatographic methods where it is clinically necessary is still important. This decision however is different to the use of the GCMS for every urine drug sample screening.
Conclusion
The pathology initiated change for improving TAT of DOA testing has resulted in the provision of a rapid and relatively inexpensive service for most of the patients across our network to aid critical clinical care decisions. The change led to almost instantaneous change in test ordering behaviour by clinicians without leading to anyobvious clinical harm, to a very significant improvement in TAT and cost savings, immediately and then sustained.


