Influence of age and gender variations on glomerular filtration rate estimated by the MCQE formula
Giuseppe Lippi
[*]
[1]
Nicola Tessitore
[2]
Martina Montagnana
[1]
Valeria Bedogna
[2]
Gian Luca Salvagno
[1]
Giovanni Targher
[3]
Antonio Lupo
[2]
Gian Cesare Guidi
[1]
Introduction
Glomerular filtration rate (GFR) is considered the best index of kidney function, which is also recommended by the National Kidney Foundation (NKF), the Kidney Disease Outcomes Quality Initiative (K/DOQI) and the National Kidney Disease Education Program (NKDEP) for diagnosing renal damage and classifying the severity of kidney disease (1). Traditionally, GFR cannot be measured by direct means, but it can be assessed by measuring urinary clearance of exogenous filtration markers. However, due to difficulty in use, expenses, radiation exposure, and radionuclide regulatory requirements, these methods have limited use and are typically confined to the research setting (2). Therefore, it is now recommended to estimate GFR by equations based on serum creatinine, either to assess renal function (2)or to predict cardiovascular outcome (3). Several formulas have been proposed so far, but the currently recommended equation is that developed and validated from the Modification of Diet in Renal Disease (MDRD) Study (2). This formula is superior to the traditional Cockcroft-Gault equation for prediction of radionuclide determined GFR of < 60 mL/min/1.73 m2 and is more accurate when creatinine results lower than 60 µmol/L are either corrected to 60 µmol/L or excluded from GFR calculations (4). However, it has also been highlighted that this formula might have some drawbacks, since it fails to correct results for age and gender (5), and it has not been sufficiently tested in children, the elderly, pregnant women, patients with serious comorbidities, or persons with extremes of body size (1). Moreover, since it has also been demonstrated that the MDRD formula might significantly underestimate the rate of decline in GFR when measured by a reference method, the new Mayo Clinic Quadratic Equation (MCQE) has been developed (6). Therefore, the main purpose of this study was to assess the influence of age and gender variations on GFR estimated by this new equation.
Patients and methods
Patients
Results of serum creatinine tests, which were performed in consecutive outpatients referred by general practitioners for routine blood testing over the past year, were retrieved from the database of our Laboratory Information System at the University Hospital of Verona. Fasting venous blood was routinely collected from outpatients in the morning.
Methods
Serum creatinine was measured on a Roche/Hitachi Modular System P (Roche Diagnostics GmbH, Mannheim, Germany) by creatinine Jaffe, rate blanked and compensated assay. The quality of results throughout the study was validated through regular internal quality control procedures and participation in an External Quality Assessment Scheme. GFR was estimated on the entire study population by the MDRD (2):
GFR = 186 x (serum creatinine–1.154) × (age–0.203) × 1.212 (if African-American) × 0.742 (if female);
and MCQE:
GFR = exp [1.911 + (5.249/serum creatinine) – (2.114/serum creatinine2) – 0.00686 × age– 0.205 (if female)].
If serum creatinine value is < 71 μmol/L it is replaced by 71 μmol/L (6).
Statistical analysis
The Shapiro-Wilkinson test was used for assessment of normality of variable distributions. Variables were logarithmically transformed to improve normality prior to analysis. The Kruskal-Wallis test was used to evaluate the existence of statistically significant difference in GFR according to age decades. Statistical analyses were performed using the statistical package SPSS version 12.0 (SPSS, Chicago, IL) and the level of statistical significance was set at 0.05. Data are presented as geometric mean and 95% confidence interval (CI).
Results
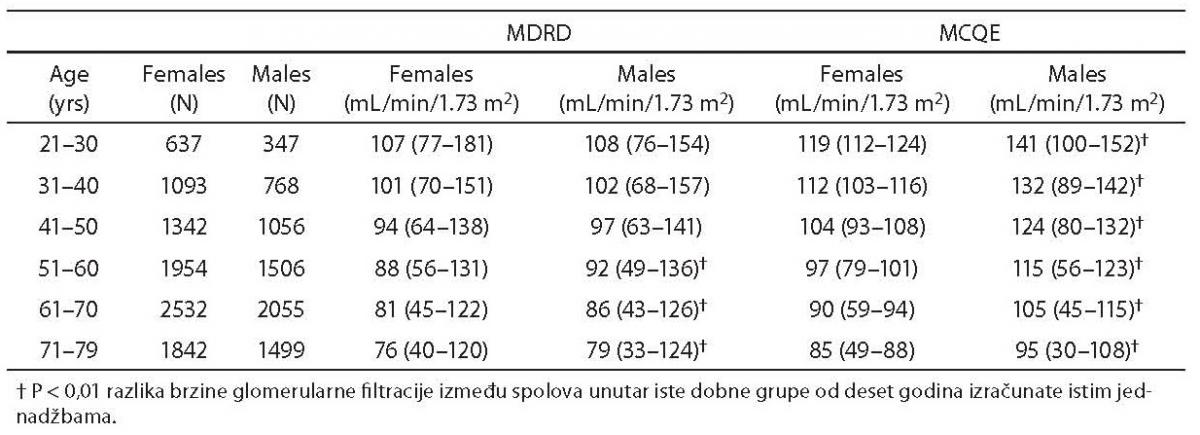
Cumulative results for serum creatinine levels were retrieved for 16,631 outpatients over the 1-year period (M/F=7,231/9,400; age 57±15 years; range 21–79 years). The mean values (95% CI) of creatinine, MDRD- and MCQE-estimated GFR were 72 μmol/L (48–128 μmol/L), 78 mL/min/1.73 m2 (38–129 mL/min/1.73 m2) and 94 mL/min/1.73 m2 (35–134 mL/min/1.73 m2), respectively. Gender difference in the estimated GFR was significantly different when calculated with either the MDRD [mean (95% CI)]: [91 (44–127) vs. 87 (48–138) mL/min/1.73 m2; P < 0.001] or MCQE [109 (46–142) vs. 95 (65–120) mL/min/1.73 m2; P < 0.001] formula. Significant gender differences were observed in all age decades by MCQE and in subjects older than 51 by MDRD (Table 1). A significant difference in the mean GFR values estimated by either formula was also observed between different age decades in both genders (P < 0.001 all). Linear regression analysis revealed an inverse association between age and estimated GFR by both the MDRD (males: standardized beta coefficient = –0.384; P < 0.001; females: standardized beta coefficient = –0.437; P < 0.001) and MCQE (males: standardized beta coefficient = –0.639; P < 0.001; females: standardized beta coefficient = –0.816; P < 0.001) formulas, with a constant mean decrease of 7% for each decade increase in age, in both genders and for either equation.
Table 1. Values of glomerular filtration rate (geometric mean and 95% confidence interval) estimated by Modification of Diet in Renal Disease (MDRD) or Mayo Clinic Quadratic Equation (MCQE) formulas and stratified according to age and sex
Discussion
Chronic kidney disease is a major public health problem worldwide, whose incidence and prevalence have more than quadrupled over the last 2 decades (1,2). GFR is considered the best overall index of kidney function for diagnosing, staging, and appropriately treating kidney disease. Due to well-recognized limitations of serum creatinine concentration as an index of kidney function, international organizations now recommend the use of GFR estimating equations based on serum creatinine and other demographic and clinical variables, and that clinical laboratories report estimated GFR whenever serum creatinine is ordered (7). Accordingly, laboratory professionals must clearly acknowledge the importance of a reliable estimation of GFR to improve both the diagnosis and treatment of patients. Although calculation of GFR by the MDRD equation is widely used, since the formula is derived from the urinary clearance of 125I-iothalamate on a large sample size where gender differences in creatinine levels have been taken into consideration, it has been demonstrated that the recently developed MCQE formula might improve prediction of GFR, especially in diabetic subjects (8). Current reports suggest the same cut-off (60 mL/min 1.73 m2) of GFR when estimated by MDRD and MCQE formulas in both genders. Recently, Khatami et al. have highlighted that a univocal threshold for MDRD derived GFR cannot be used throughout different genders and ages (9), since the difference between the men and women is significantly different and there is an inverse association between age and GFR in both genders. However, no information is available on the potential influence of age and gender variations on the MCQE derived GFR. Although it has recently been hypothesized that GFR estimation by this newly developed formula might be a more accurate approach (6),the results of this epidemiological analysis confirm that even the MCQE equation might fail to compensate for age and gender variations, requiring implementation of different corrective actions. This is of particular concern, in that the difference between genders and age decades may be due to the fact that smaller body size has lower metabolic demands and shorter individuals require less renal function. As a logical consequence, the lack of age- and gender-adjusted reference ranges for both MDRD and MCQE estimates implies that many patients with moderate to severe renal failure might go misdiagnosed.
We are aware that the outcome of this investigation might have some limitations. In fact, the retrospective audit is only representative of the population served by our laboratory, and we lack clinical information on request forms and one-point follow up analysis. It will therefore require further prospective investigations to confirm these preliminary findings, preferably in more heterogeneous population settings. Moreover, we could not assess GFR by the gold standard approach. However, it is to mention that GFR calculation by the current reference approach that employs the clearance of exogenous filtration markers is discouraged in populations of healthy subjects, and the use of cystatin C-derived equations is superior to creatinine-based prediction equations only in children aged < 14 years (2).
Notes
Potential conflict of interest
None declared.
References
1. National Kidney Foundation. K/DOQI clinical practice guidelines for chronic kidney disease: evaluation, classification, and stratification. Kidney Disease Outcome Quality Initiative. Am J Kidney Dis 2002;39:S1-S246.
2. Myers GL, Miller WG, Coresh J, Fleming J, Greenberg N, Greene T, et al. National Kidney Disease Education Program Laboratory Working Group. Recommendations for improving serum creatinine measurement: a report from the Laboratory Working Group of the National Kidney Disease Education Program. Clin Chem 2006;52:5-18.
3. Ruilope LM, Zanchetti A, Julius S, McInnes GT, Segura J, Stolt P, et al. VALUE Investigators. Prediction of cardiovascular outcome by estimated glomerular filtration rate and estimated creatinine clearance in the high-risk hypertension population of the VALUE trial. J Hypertens 2007;25:1473-9.
4. Saleem M, Florkowski CM, George PM, Woltersdorf WW. Comparison of two prediction equations with radionuclide glomerular filtration rate: validation in routine use. Ann Clin Biochem 2006;43:309-13.
5. Khatami Z, Handley G, Narayanan K, Weaver JU. Applicability of estimated glomerular filtration rate in stratifying chronic kidney disease. Scand J Clin Lab Invest 2007;67:297-305.
6. Rule AD, Larson TS, Bergstralh EJ, Slezak JM, Jacobsen SJ, Cosio FG. Using serum creatinine to estimate glomerular filtration rate: accuracy in good health and in chronic kidney disease. Ann Intern Med 2004;141:929-37.
7. Chi-YuanHsu, Chertow GM, Curhan GC. Methodological issues in studying the epidemiology of mild to moderate chronic renal insufficiency. Kidney Int 2002;61:1567-76.
8. Rigalleau V, Lasseur C, Raffaitin C, Perlemoine C, Barthe N, Chauveau P, et al. The Mayo Clinic quadratic equation improves the prediction of glomerular filtration rate in diabetic subjects. Nephrol Dial Transplant 2007;22:813-8.
9. Khatami Z, Handley G, Narayanan K, Weaver JU. Applicability of estimated glomerular filtration rate in stratifying chronic kidney disease. Scand J Clin Lab Invest 2007;67:297-305.