Bisalbuminemia in a male Croatian patient with sarcoidosis
Ana-Maria Simundic
[*]
[1]
Marijana Miler
[1]
Nora Nikolac
[1]
Elizabeta Topić
[1]
Dubravka Čaržavec
[2]
Branka Milanović
[1]
Vladimir Stančić
[2]
Introduction
Bisalbuminemia represents a rarely encountered abnormality, characterised by the occurrence of bicuspid electrophoretic pattern in the albumin fraction detected on serum electrophoresis (1). This albumin abnormality may be both genetic and acquired. Serum albumin is non-glycosylated, negatively charged, most abundant plasma protein and its mutations and polymorphisms have recently been extensively reviewed by Minchiotti et al. (2). So far, there are 77 known mutations of the serum albumin gene, with 65 of them resultingin bisalbuminemia and 12 in analbuminemia (state of serum albumin concentration less than 1 g/L). Inherited bisalbuminemia is an autosomal dominant condition (3) which occurs with the cumulative frequency of 1:1000 to 1:10,000 (4). Acquired forms of bisalbuminemia are described in patients receiving high penicillin doses (5,6) and in patients with ascites or pancreatic pseudocyst (7).
Though being mostly benign, bisalbuminemia has been described in several pathological conditions such as nephrotic syndrome (8,9), chronic kidney disease (10), Alzheimer disease (11) and benign monoclonal gammopathy (12). Two genetic variants have already been described in two Croatian families i.e. slow-migrating albumin Zagreb and fast-migrating albumin Krapina (13,14).
In this article we report a case of bisalbuminemia in a 46-year-old Croatian male patient with sarcoidosis. To the best of our knowledge, bisalbuminemia in sarcoidosis has not yet been reported.
Materials and methods
Case history
A 46-year-old non-smoking male Croatian (M. L.) was referred by a general practitioner to the University Department of Internal Medicine, Sestre milosrdnice University Hospital (Zagreb, Croatia) for a diagnostic work-up with symptoms of erythema nodosumof lower extremities and bilateral hilar lymphadenopathy. The patient was afebrile, denied fatigue and any respiratory symptoms. His past medical history included hyperlipidemia for the last 15 years and excessive alcohol consumption (1 liter of wine daily) for the last two years. The patient claimed he had received a three-day prophylactic course of azythromicin during the previous week for diseases transmitted by insect bite. Prior to his visit to our Department, the patient had during a two-week period undergone some examinations elsewhere and obtained the following results: bone scintigraphic examination of the lower leg showed no abnormalities, radiograph of the lower leg showed osteoid osteoma in the left tibia, chest radiograph revealed enlarged bilateral pulmonary hila, urine culture was negative, kidney ultrasound showed two small cysts on the left kidney. Red blood cell count (RBC), white blood cell count (WBC), C-reactive protein, uric acid, rheumathoid factor were also within referent interval. The patient was of normal body weight and otherwise well. He was admitted to our Department for further diagnostic work-up. Written consent was obtained from the patient to publish this report.
Methods
Our standard diagnostic work-up for suspected sarcoidosis included: standard 12-lead electrocardiogram (EKG), chest X-ray radiography. The patient was seen by a pulmonologist. He underwentspirometry examination to assess the forced vital capacity (FVC), forced expiratory volume in 1 second (FEV1) and the FEV1/FVC ratio.
Urine, serum and plasma (EDTA and citrate) were collected for routine laboratory tests. Biochemistry parameters were assayed on automated Olympus AU2700 clinical chemistryanalyzer (Olympus, Hamburg, Germany). Haematology and coagulation parameters were determined with automated analyzers Cell-Dyn Sapphire (Abbott Laboratories, Abbott Park, IL, USA) and BCS Coagulation Analyzer (Dade Behring, Marburg, Germany), respectively.
Immunoglobulin IgA, IgG and IgM concentrations were determined by nephelometry using the automated BN II analyzer (Dade Behring, Marburg, Germany).
Serum capillary protein electrophoresis was performed with automated capillary electrophoresis Capillarys System (Sebia, Issy-les-Moulinaux, France) according to manufacturer’s instruction for Capillarys protein(E) 6 kit (Sebia, Issy-les-Moulinaux, France). Briefly, the samples, automatically diluted with buffer (1:10), were injected at the anodic end of silica capillaries. Charged molecules were separated by their electrophoretic mobility in an alkaline buffer (pH 9.9) at 300 V. Protein separation was performed in capillaries and direct detection of proteins was made at 200 nm at the cathodic end of the capillary.
Diagnosis of sarcoidosis was made based on patient history, compatible clinical features along with laboratory and pathologic findings (15).
Results
Medical examination revealed the following: patient chest radiography confirmed previously observed enlarged bilateral pulmonary hila, without any interstitial changes. Spirometry revealed normal ventilation: forced expiratory volume in one second (FEV1) of 4.94l (118%), and forced vital capacity (FVC) of 5.22l (115%); FEV1/FVC ratio was 0.95.
Haematological, coagulation and most of the biochemical parameters were within the normal range with the exception of total cholesterol (7.7 mmol/L; recommended value <5.0 mmol/L), LDL-cholesterol concentration (5.3 mmol/L; recommended value <3.0 mmol/L) and angiotensin-converting enzyme (ACE) activity (64.5 U/L; referent range <52 U/L)which were elevated. The patient had serum IgM, IgA and IgG immunoglobulin concentrations within the normal range.
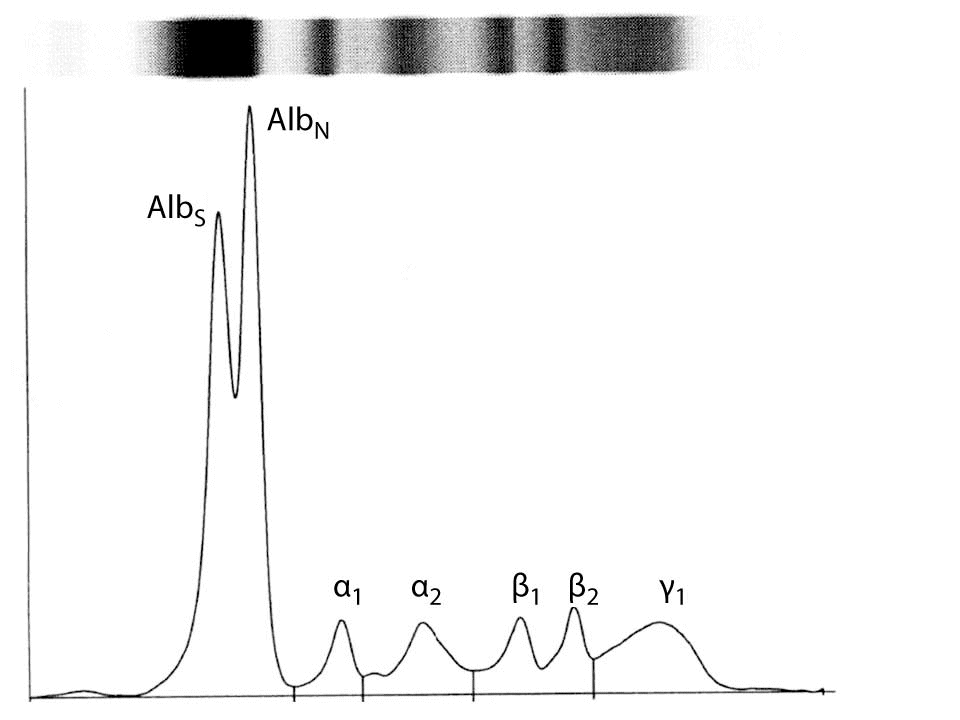
Serum electrophoresis revealed two distinct albumin bands at anodic end (Figure 1). The bands were of unequal relative amounts. New albumin variant band had slower mobility compared to normal albumin. Total protein concentration was 65 g/L, with the albumin to globulin ratio of 1.63 (reference range: 0.80–2.00). Slow-migrating and normal band concentrations were 0.48 and 0.51 of the total albumin, respectively. The patient’s serum was re-examined and remained bisalbuminemic three and six months after his first visit.
The patient’s first degree relative (sister, D. L.) also consented to give blood sample. Her serum electrophoretic pattern was identical to our bisalbuminemic patient (M. L.), which indicated the inheritable nature of the albumin variant.
Figure 1. Capillary electrophoresis of serum proteins from a patient with sarcoidosis. Serum proteins are separated into following fractions: AlbS (newly encountered slow-migrating albumin variant), AlbN (normal albumin), α1, α2, β1, β2 and γ globulins.
Discussion
Bisalbuminemia was observed by several authors more than 50 years ago (1,16,17) and it has been under intense investigation by several research groups ever since. So far, vast majority of albumin molecular defects have been extensively characterized (2). Those data have provided solid basis for further studies on the ligand binding properties of the albumin molecule (14,18,19). Although variants with altered affinity to triiodothyronine (T3) and thyroxine (T4) (20), fatty-acids (21) have been described, the overall effect of the amino-acid sequence change on the ligand-binding properties is supposed to be negligible and of minor clinical significance. Furthermore, bisalbuminemia has so far most commonly been reported as benign condition, found as a concomitant phenomenon, secondary to the underlying disease or pathological condition. The only exception is familial dysalbuminemic hypertriiodothyroninemia, first reported by Sunthornthepvarakul et al. (22) in a clinically euthyroid members of a Thai family that presented with high serum total T3 concentration. Further evidence on the role of albumin variant as the underlying cause of hypertriiodothyroninemia was provided by Nakamura et al. (23) in a report on a 56-year-old Japanese female. Clinical endocrinologists should always consider the presence of possible albumin variant in a patient with elevated T3 and T4 when there are no other obvious reasons for this increase. Capillary zone electrophoresis (CZE) has been shown by some authors to be superior to agarose gel electrophoresis because of the substantially improved separation of albumin, α1 and α2 fractions (12,24). Therefore, when the presence of albumin variant is suspected, CZE should be performed since it is the electrophoretic method of choice for this purpose.
In this study, we present a new case of inherited bisalbuminemia in a patient with sarcoidosis. To the best of our knowledge, bisalbuminemia has never been previously reported in sarcoidosis. Sarcoidosis is a multisystem disease of unknown aetiology with multiple clinical manifestations and pathological findings. Whether the occurrence of bisalbuminemia has any relevance to the etiopathophysiologicalmechanism of this disease remains to be elucidated. At this moment we were unfortunately not able to further explore the nucleic acid sequence as well as the structural and functional characteristics of this albumin variant. It is our aim to perform such analysis in collaboration with some other research groups.
Both clinical chemists and medical doctors should always be alerted by every new case of either inherited or acquired albumin variant because it could provide some new data on the protein evolution as well as the physical, chemical and molecular characteristics of the albumin molecule, which may lead to the discoveryand development of some new potential therapeutic approaches. If proven to be acquired, bisalbuminemia finding may point to some underlying pathophysiologicalcondition such as pancreatic pseudocysts, offering the physician some additional clinical information which may help in differential diagnosis.
Acknowledgement
This study was supported by the Ministry of Science, Education and Sports, Republic of Croatia; project number: 134-1340227-0200. The authors would also like to thank to Prof. Slavica Dodig, PhD (Srebrnjak Children’s Hospital), for very useful comments and kind assistance in preparing this manuscript.
Notes
Potential conflict of interest
None declared.
References
1. Sarcione EJ, Aungst W. Studies in bisalbuminemia: Binding properties of the two albumins. Blood 1962;20:156-64.
2. Minchiotti L, Galliano M, Kragh-Hansen U, Peters T Jr. Mutations and polymorphisms of the gene of the major human blood protein, serum albumin. Hum Mutat 2008;29:1007-16.
3. Kumit DM, Phillip BW, Bruns GA. Confirmation of the mapping assignment of human serum albumin to chromosome 4 using a cloned human albumin gene. Cytogenet Cell Genet 1982;34:282-8.
4. Tárnoky AL. Genetic and drug-induced variation in serum albumin. Adv Clin Chem 1980;21:101-46.
5. Arvan DA, Blumberg BS, Melartin L. Transient “bisalbuminemia” induced by drugs. Clin Chim Acta 1968;22:211-8.
6. Rocha J, Bohner J, Kömpf J. Transient bisalbuminemia: separation by isoelectric focusing of human albumin fractions linked to different numbers of benzylpenicilloyl groups. Electrophoresis 1995;16:1031-3.
7. Kobayashi S, Okamura N, Kamoi K, Sugita O. Bisalbumin (fast and slow type) induced by human pancreatic juice. Ann Clin Biochem 1995;32:63-7.
8. Hoang MP, Baskin LB, Wians FH Jr. Bisalbuminuria in an adult with bisalbuminemia and nephrotic syndrome. Clin Chim Acta 1999;284:101-7.
9. Ahmad J, Khan AS, Siddiqui MA, Tewari SG, Khan RU. Bisalbuminemia in nephrotic syndrome (a case report). Jpn J Med 1984;23:45-7.
10. Ejaz AA, Krishna M, Wasiluk A, Knight JD. Bisalbuminemia in chronic kidney disease. Clin Exp Nephrol 2004;8:270-3.
11. Shetty JK, Prakash M, Gopalakrishna K. Bisalbuminemia in an adult male with Alzheimer’s disease. Indian J Med Sci 2007;61:356-7.
12. Kalambokis G, Kitsanou M, Kalogera C, Kolios G, Seferiadis K, Tsianos E. Inherited bisalbuminemia with benign monoclonal gammopathy detected by capillary but not agarose gel electrophoresis. Clin Chem 2002;48:2076-7.
13. Dodig S, Cepelak I, Benko B, Raos M, Branovic K. Bisalbuminemia in two Croatian families. Arch Med Res 2000;31:608-12.
14. Kragh-Hansen U, Campagnoli M, Dodig S, Nielsen H, Benko B, Raos M, et al. Structural analysis and fatty acid-binding properties of two Croatian variants of human serum albumin. Clin Chim Acta 2004;349: 105-12.
15. Judson MA. Sarcoidosis: clinical presentation, diagnosis, and approach to treatment. Am J Med Sci 2008;335:26-33.
16. Knedel M. Double albuminemia, a new hereditary protein anomaly. Blut 1957;3:129-34.
17. Wuhrmann F. Double albumin peak as a hereditary blood protein anomaly. Schweiz Med Wochenschr 1959;89:150-2.
18. Reed RG. Ligand-binding properties of albumin Parklands: Asp365----His. Biochim Biophys Acta 1988;965:114-7.
19. Kragh-Hansen U, Chuang VT, Otagiri M. Practical aspects of the ligand-binding and enzymatic properties of human serum albumin. Biol Pharm Bull 2002;25:695-704.
20. Sarcione EJ, Aungst CW. Bisalbuminemia associated with albumin thyroxine-binding defect. Clin Chim Acta 1962;7:297-8.
21. Minciotti L, Kragh-Hansen U, Nielsen H, Hardy E, Mercier A-Y, Galliano M. Structural characterization, stability and fatty acid-binding properties of two French genetic variants of human serum albumin. Biochim Biophys Acta 1999;1431:223-31.
22. Sunthornthepvarakul T, Likitmaskul S, Ngowngarmratana S, Angsusingha K, Kitvitayasak S, Scherberg NH, et al. Familial dysalbuminemic hypertriiodothyroninemia: a new, dominantly inherited albumin defect. J Clin Endocrinol Metab 1998;83:1448-54.
23. Nakamura S, Kajita Y, Ochi Y. Familial dysalbuminemic hypertriiodothyroninemia in a Japanese kindred. Intern Med 2000;39:50-4.
24. Jaeggi-Groisman SE, Byland C, Gerber H. Improved sensitivity of capillary electrophoresis for detection of bisalbuminemia. Clin Chem 2000;46:882-3.