Introduction
Chronic obstructive pulmonary disease (COPD), one of the leading health problems in developed countries, is a multisystemic disease that starts with lung morphology and function impairment and progresses to multiple systemic manifestations such as systemic inflammation processes, immunodeficiency, frequent respiratory infections, malnutrition, osteoporosis, cardiovascular complications (1) and, as recently reported, to anemia (2).
Continuous scientific and professional interest in COPD is therefore justified and aimed at prevention and early detection of the disease, as well as at search for the most efficient treatment, improvement of the quality of life, and prolonged survival. Inflammation, characterized by an increased neutrophil count, is one of the key factors in the pathogenesis of this disease. Inflammatory response starts with activation of alveolar macrophages, which synthesize neutrophil chemoattractants, leukotriene (LTB4) and interleukin-8 (IL-8). They further activate neutrophils, the most important effector cells in inflammatory events in COPD. Namely, activated neutrophils synthesize and excrete proteolytic enzymes (e.g., neutrophilic elastase, cathepsins and matrix metalloproteinases) and reactive oxygen molecules that directly damage lung tissue (3). The concentration of acute phase proteins is also elevated in the blood of COPD patients. Literature data point to raised C-reactive protein (CRP) concentration as a marker of systemic inflammation in these patients (4).
Owing to accompanying hypoxia, COPD is almost traditionally associated with polycythemia (5). Besides polycythemia, anemia also occurs in COPD patients; according to literature, its incidence is 5%-33% (6). It is assumed that the mechanism of anemia development in COPD is the same as that of anemia of inflammation (7). Namely, elevated concentrations of inflammatory cytokines (IL-1, TNF-alpha, IFN-gamma, etc.) lead to a shortening of erythrocyte life, disturbed utilization of iron from reticuloendothelial stores, and to impairment of the compensational activity of bone marrow. Hence, bone marrow does not respond to enhanced erythropoietin (EPO) formation, resistance to EPO emerges, and anemia develops, most commonly normocytic and normochromic, seldom microcytic and hypochromic anemia (8). It is assumed that anemia could be a significant prognostic factor in COPD patients, because decreased hemoglobin and hematocrit concentrations in these patients are associated with frequent and long-term hospitalization and increased mortality (9).
The goals of this study were: (a) to establish the relative proportion of anemia in all COPD patients and differences in hematological markers of anemia between COPD patients and patient subgroups according to The Global Initiative for Chronic Obstructive Lung Disease (GOLD) guidelines in comparison with control group; (b) to establish differences in CRP concentration as a marker of systemic inflammation between all COPD patients and patient subgroups according to the GOLD guidelines in comparison with control group; (c) to establish differences in CRP concentration between the subgroups of patients with and without anemia; (d) to assess the possible correlation between CRP concentration and selected hematological markers of anemia; and (e) to determine diagnostic accuracy of CRP in discriminating between patients with COPD and healthy subjects.
Materials and methods
Subjects
The study included 150 COPD patients and 51 control subjects. In the group of COPD patients there were 105 (70%) male and 45 (30%) female patients, median age 71 (63 and 76, respectively). All patients were treated at Department of Pulmonary Diseases, Dr. Ivo Pedišić General Hospital, Sisak, during the period from May 2007 to June 2008. COPD was diagnosed on the basis of history data, clinical findings, and results of functional spirometric tests. Patients with other pulmonary diseases, acute inflammatory diseases, liver diseases, diabetes mellitus and cancer history were excluded from the study. Based on the stage of the disease, i.e. spirometry result, considered as the “gold standard“ in diagnosing this disease, patients were allocated to four subgroups according to the GOLD guidelines: I, IIA, IIB, and III (GOLD I mild form, FEV1 (forced expiratory volume in 1 sec) ≥ 80% of anticipated value; GOLD II A moderate form, 50% ≤ FEV1 < 80% of anticipated value; GOLD II B moderate form, 30% ≤ FEV1 < 50% of anticipated value; and GOLD III severe form, FEV1 < 30% of anticipated value or FEV1 < 50% of anticipated value with respiratory failure or with clinical signs of right heart failure (10).
GOLD I subgroup comprised 17 (11%), subgroup IIA 47 (31%), subgroup IIB 50 (33%) and subgroup III 36 (24%) patients. Study patients were further divided into two subgroups according to the World Health Organization (WHO) criteria for anemia: hematocrit value < 0.390 L/L for males, and < 0.360 L/L for females (11). Subgroup A (with anemia) included 36 (36/150 = 24%) patients, and subgroup B (without anemia) 114 (114/150 = 76%) patients. Control group included 51 subjects, 21 (41%) males and 30 (59%) females, median age 52 (46 and 56, respectively). They were selected among the individuals presenting for regular medical check up at Sisak and Petrinja Health Centers, and were without any signs of acute or chronic condition. All volunteers were subjected to the standard pulmonary function test as the main criterion for inclusion in the study. Namely, volunteers were included in the study as controls on the basis of normal spirometric finding.
All patients and control subjects gave a written consent to be included in the study, which was approved by the Ethics Committee of Dr. Ivo Pedišić General Hospital.
Samples
Whole blood samples collected with K2EDTA as anticoagulant were used for hematologic tests, i.e. erythrocytes, hemoglobin, hematocrit, red blood cell constants: mean cell volume of red cells (MCV), mean cell hemoglobin content of red cells (MCH), mean cell hemoglobin concentration (MCHC), and reticulocytes.
CRP concentration was determined in sera obtained by centrifugation at 1300 x g for 10 minutes in test tubes without anticoagulant.
Methods
Hematological tests were done according to standard procedures based on flow cytometry on an automatic Cell Dyn 3200 CS cell counter (Abbott Diagnostics). Reticulocyte count was determined with the same device after staining with New Methylene Blue N. CRP concentration was determined by the immunoturbidimetric method on an automated Dimension Xpand Plus analyzer (Siemens Healthcare Diagnostics) using test reagents of the same manufacturer.
Statistical analysis
Each set of data were analyzed for normality by the Kolmogorov-Smirnov statistical test. Normally distributed data were expressed as arithmetic mean ± standard deviation (x ± SD), while the non-normally distributed data were expressed as median and interquartile range. Significance of between-group differences was tested by the parametric independent t-test and nonparametric Mann-Whitney test.
To compare all study analytes among the four GOLD subgroups, the one-way analysis of variance (ANOVA) and nonparametric Kruskal-Wallis test were used. Statistical significance was set at P < 0.05.
To determine the correlation between CRP concentration and hematologic indicators of anemia (hemoglobin, hematocrit, reticulocytes) in the group of COPD patients and in the subgroups, Pearson’s and Spearman’s coefficients of correlation were calculated. The Receiver Operating Characteristic (ROC) analysis was performed to estimate diagnostic accuracy of CRP in discriminating between COPD patients and control group. Statistical analyses were performed using the Med Calc 9.6.0.0 for Windows 98 software.
Results
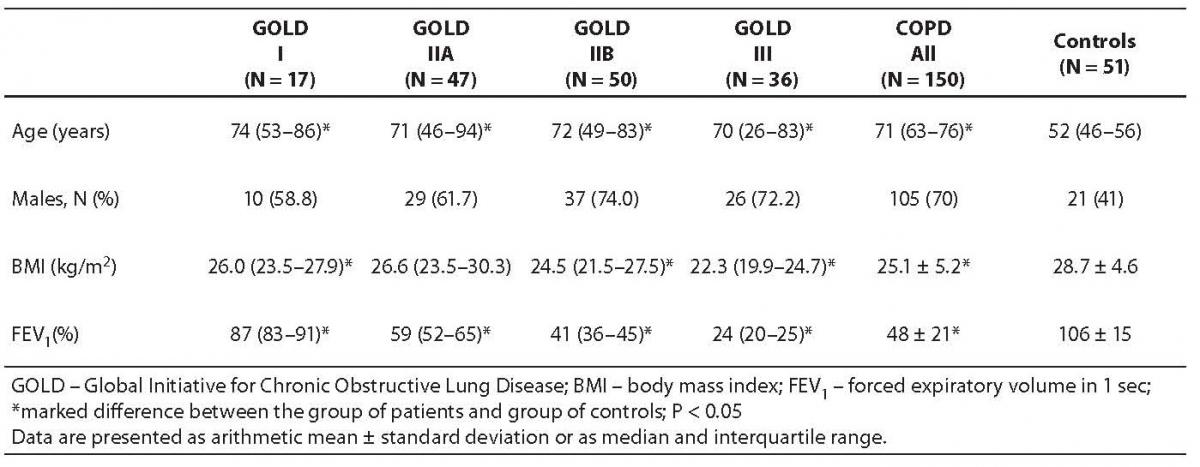
Baseline demographic characteristics of the group of COPD patients, patient subgroups according to GOLD and control group are presented in Table 1.
Table 1. Baseline demographic characteristics of all patients with chronic obstructive pulmonary disease (COPD), subgroups of patients according to GOLD classification and control subjects
In the group of COPD patients, the values of body mass index (BMI) differed significantly (P < 0.001) from the values recorded in control subjects. The difference was also statistically significant when GOLD subgroups I, IIB and III were compared with control group (P = 0.027, P = 0.001 and P < 0.001, respectively).
The groups of COPD patients and control subjects were not age matched the latter being significantly younger from both all COPD patients and GOLD subgroups (P < 0.001). FEV1 values yielded significant difference between the group of all COPD patients, GOLD subgroups of patients and control group (P < 0.001).
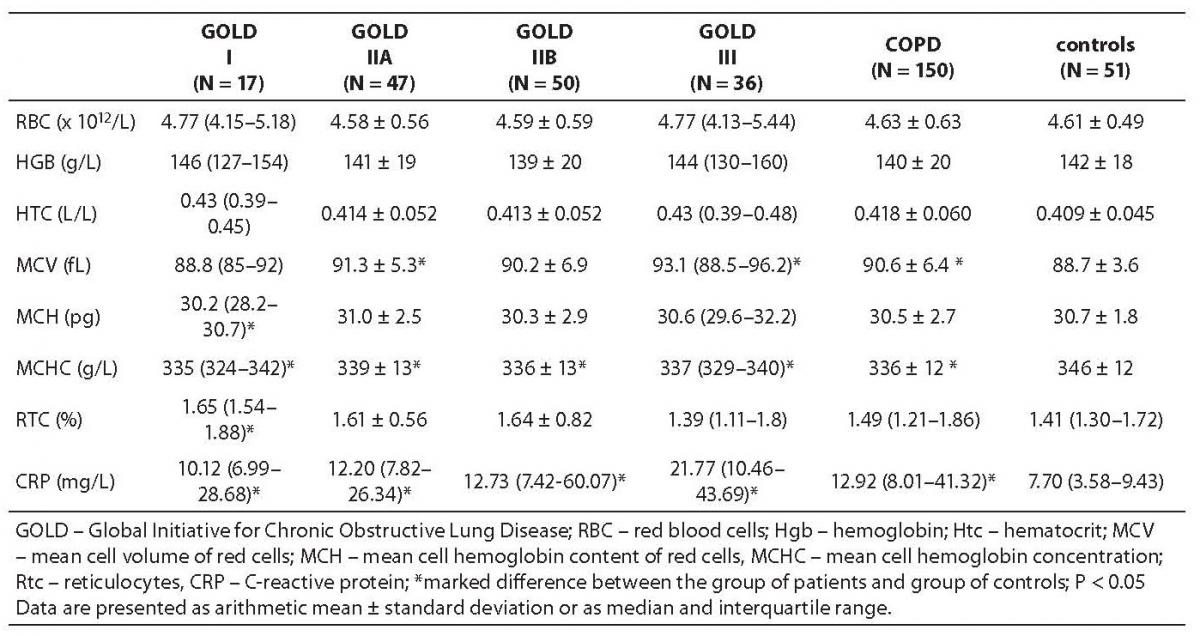
The values of hematological indicators and CRP in the group of all COPD patients, GOLD subgroups and control group are presented in Table 2.
Table 2. Values of hematologic markers and CRP concentration in patients with chronic obstructive pulmonary disease (COPD), COPD patients distributed in GOLD subgroups, and control subjects
The values of erythrocyte constants MCV and MCHC differed significantly between the group of COPD patients and control subjects (P = 0.012 and P < 0.001, respectively). The median of CRP concentration was by about 50% higher in the group of COPD patients, yielding a significant difference from the control group (P < 0.001).
The values of erythrocytes, hemoglobin and hematocrit did not differ significantly between any of the GOLD patient subgroups and control subjects.
MCV value was significantly higher in the GOLD IIA subgroup (P < 0.008) and MCH in the GOLD I subgroup (P < 0.039) only, whereas MCHC was significantly lower in all GOLD subgroups as compared to control group (P < 0.001, P = 0.018, P < 0.001 and P < 0.001, respectively). Thee median CRP concentration increased from GOLD I subgroup towards GOLD III subgroup, where it was 21.77 mg/L or threefold median CRP concentration recorded in control group (subgroup I, P < 0.001; IIA, P < 0.001; and IIB and III, P < 0.001).
Partial oxygen pressure (pO2) in COPD patients (9.38 ± 1.93 kPa) was significantly lower as compared to the reference values (10.00-13.33 kPa). COPD patients also showed an increase in leukocyte count (10.24 ± 4.21 x 109/L) (results not shown).
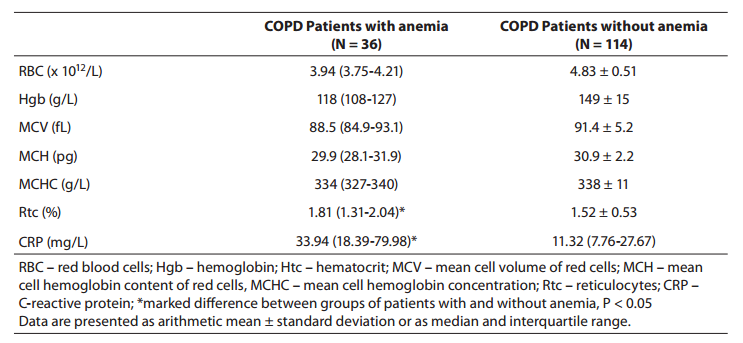
The values of hematological markers and CRP in COPD patients divided into subgroups according to the WHO criteria for anemia are presented in Table 3. Anemia was present in 36 (24%) patients.
Table 3. Values of hematologic markers and CRP concentrations in patients with chronic obstructive pulmonary disease (COPD) distributed in groups according to World Health Organization criteria for anemia
In group A patients (with anemia), reticulocyte count was significantly higher (P = 0.003), while median CRP concentration was threefold that recorded in group B patients (without anemia) (P < 0.001).
Comparison of the study analytes in all four GOLD subgroups showed no statistically significant difference.
Determination of the coefficient of correlation between hemoglobin concentration and CRP and between hematocrit and CRP in the group of COPD patients produced a weak negative correlation (r = -0.28; P < 0.001 and r = -0.27; P = 0.001, respectively) (12), while there was no correlation between reticulocyte count and CRP concentration. The strongest negative correlation was obtained by determination of the correlation coefficient between hemoglobin concentration and CRP (r = -0.60; P < 0.001), and between hematocrit and CRP (r = -0.59; P < 0.001) (12) in the GOLD III subgroup.
Although CRP concentration was significantly higher in patients with anemia than in those without it, there was no correlation between CRP concentration and hemoglobin or hematocrit, and no correlation between CRP concentration and reticulocyte count either in the group of patients with anemia.
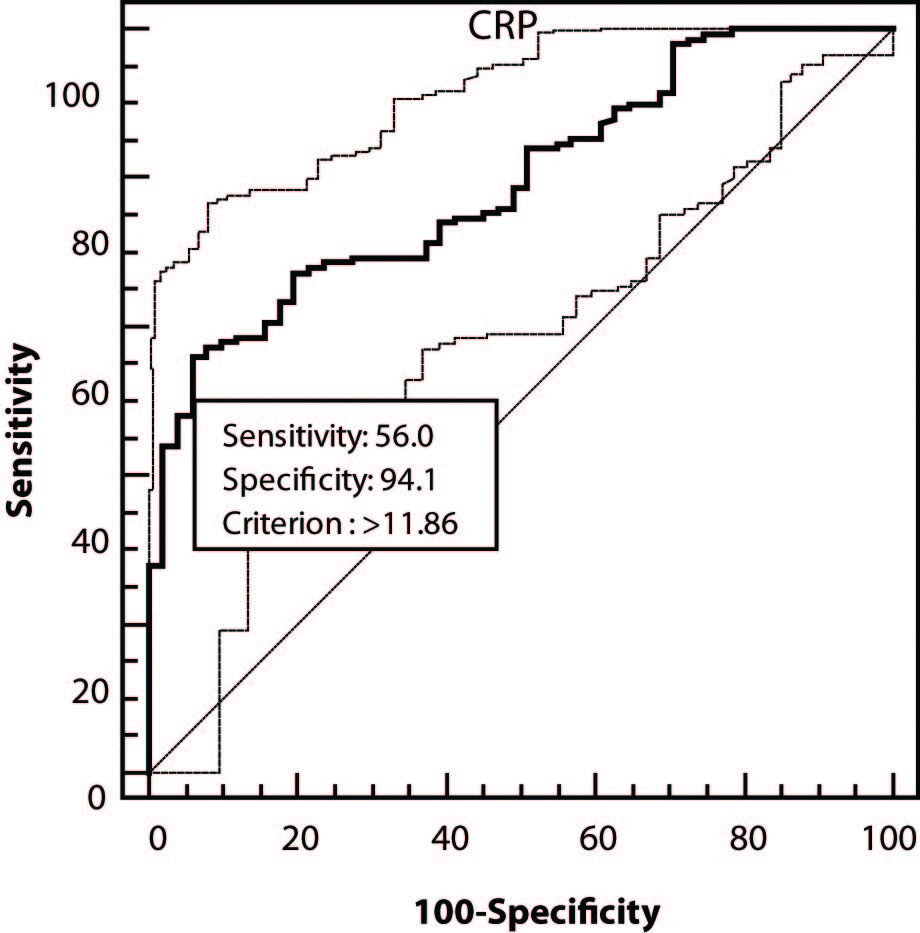
Good diagnostic efficiency of CRP was determined by ROC analysis, comparing the group of all COPD patients with control group (area under the curve, AUC = 0.795; 95%CI = 0.732-0.849; P < 0.001; at a cut-off concentration of 11.86 mg/L, diagnostic sensitivity of 56% and specificity of 94.1% were determined) (Figure 1).
Figure 1. ROC analysis of C-reactive protein (CRP) in the group of all patients with chronic obstructive pulmonary disease.
ROC analysis was also done for all hematological indicators of anemia, comparing the group of all COPD patients with control group; however, the diagnostic efficiency thus obtained was unsatisfactory for all study parameters (AUC ≤ 0.5).
Discussion
In the group of COPD patients included in the study, anemia (decreased hematocrit and hemoglobin values, increased proportion of reticulocytes, lower MCH and MCHC values along with normal or decreased MCV values) was present in 36 (24%) patients. CRP concentration was significantly higher in the group of all patients, as well as in all GOLD subgroups as compared to control group, confirming the role of CRP as a marker of systemic inflammation in this disease. CRP concentration was significantly higher in the subgroup of COPD patients with anemia as compared to the patient subgroup without anemia, pointing to the possible association of anemia with the presence of inflammation in this disease. Reticulocyte count was also higher in the subgroup of COPD patients with anemia. The results of this study also pointed to negative correlation between hematocrit and hemoglobin values and CRP concentration (a marker of inflammation), notably in GOLD subgroup III, which includes patients with the severe form of the disease with greatly reduced pulmonary function (r = -0.60, P < 0.001; r = -0.59, P < 0.001), as also reported by Richmond and Kiss (13).
Diagnostic specificity and sensitivity of CRP were determined by ROC analysis. At the cut-off value of 11.86 mg/L, diagnostic sensitivity, i.e. test ability to recognize COPD patient, was 56%, while diagnostic specificity, i.e. the probability that the test result would exclude the disease in a healthy person, was 94%. The ROC curve and area under the curve (AUC) indicated good discriminatory efficiency of CRP, i.e. its good diagnostic accuracy. Pinto-Plata et al. (14) report that CRP could be a marker of a systemic inflammatory process present in COPD patients.
The anemia found in the group of COPD patients was mainly normocytic and normochromic (83.4%), and to a lesser extent microcytic and hypochromic (16.6%) anemia, which corresponds to the picture of anemia of inflammation (15). Namely, after iron deficiency, chronic inflammatory diseases are the most common conditions associated with anemia (anemia of inflammation, anemia of chronic disease). Thus, COPD has recently been added to the list of chronic inflammatory diseases associated with anemia, along with infections, tumors, autoimmune diseases and kidney diseases (7). Similowski et al. (7) report that COPD is a disease that used to be mainly associated with polycythemia (incidence 5%-8%) and, although it may seem paradoxical, it has only recently started to be associated with anemia (16), the incidence of which is 10%-15% (6). The proportion of patients with anemia in this study was consistent with literature data, since some authors report on the incidence of anemia of up to 33% (17). Polycythemia was present in 8% of our COPD patients, which is also in accordance with the results reported by other authors (17,18).
The large French ANTADIR (Association Nationale pour le Traitement a Domicile des Insuffisants Respiratoires) study (9) showed strong correlation between hemoglobin concentration and mortality in COPD patients. In the same study, hematocrit value was defined as an independent and most important prognostic indicator of COPD patient survival. A higher survival rate was recorded in patients with polycythemia, while higher mortality was determined in the group of patients with low hematocrit values. In recent years, polycythemia is less common in these patients, which may be attributed to better medical care of these patients, as well as to the increased utilization of angiotensin-converting enzyme (ACE) inhibitors that are widely used to treat cardiac complications in COPD patients (19). It is also assumed that hemoglobin concentration in patients with COPD reflects the balance between erythropoiesis stimulation by hypoxia (most notable in GOLD subgroup III, representing the highest degree of disease progression) and its suppression due to the presence of inflammation (20). Since pO2 values in COPD patients are lower than the reference interval, along with raised concentrations of CRP and leukocytes, the results of this study comply with this assumption.
This preliminary investigation confirmed the importance of CRP concentration as a marker of systemic inflammation in COPD patients as well as the possible association of an increase in CRP concentration with disease progression. The cut-off value of 11.86 mg/L, obtained by ROC analysis, might be an additional biochemical parameter that could help discriminate between COPD patients and healthy individuals. An appreciable proportion of patients have anemia with characteristics corresponding to those of anemia of inflammation. This study was, however, limited by the small number of subjects in the subgroups and further more extensive studies in a greater number of subjects are necessary to elucidate the mechanism of anemia development, to determine the importance of anemia as a marker or mediator of pathophysiological events in COPD, and as a prognostic factor in COPD patients.
Notes
Potential conflict of interest
None declared
References
1. Gan WQ, Man SF, Senthilselvan A, Sin DD. Association between chronic obstructive pulmonary disease and systemic inflammation: a systematic review and a meta-analysis. Thorax 2004;59:574-80.
2. John M, Hoernig S, Doehner W, Okonko D.D., Witt C, Anker SD. Anemia and Inflammation in COPD. Chest 2005;127:825-9.
3. Barnes PJ. Mediators of chronic obstructive pulmonary disease. Pharmacol Rev. 2004;56:517-48.
4. Broekhuizen R, Wouters EF, Creutzberg EC, Schols AM. Raised CRP levels mark metabolic and functional impairment in advanced COPD. Thorax 2006;61:17-22.
5. Pavliša G. ŠRazina eritropoetina, angiogenetskih faktora i upalnih citokina u serumu prije i nakon korekcije teške hipoksemije u bolesnika s kroničnom opstruktivnom bolesti plućaĆ, PHD THESIS, SVEUČILIŠTE U ZAGREBU, 2006;28-32. (IN CROATIAN)
6. Wandersee K. Is anemia another systemic inflammatory manifestation of COPD. Available at: URL:http//www.copdtrends.org/article.asp?1d=67. Accessed February 5, 2009.
7. Similowski T, Agusti A, MacNee W, Schonhofer B. The potential impact of anaemia of chronic disease in COPD; Eur Respir J 2006; 27:390-6.
8. Stančić V. ŠPatogeneza anemijeĆ. In: ŠAnemije – trajni izazov dijagnostici i terapijiĆ Getaldić B, ed. Zagreb: Medicinska naklada; 2007. (in Croatian)
9. Chambellan A, Chailleux E, Similowski T. Prognostic value of the hematocrit in patients with severe COPD receiving long-term oxygen therapy. Chest 2005;128:1201-8.
10. Romain A, Pauwels A, Buist S, Peter M, Carverley A, Jenkins C, Hurd S. Global strategy for the diagnosis, management and prevention of chronic obstructive pulmonary disease. Am J Respir Crit Care Med 2001;163:1256-70.
11. Krishnan G, Grant BJ, Muti PC, Mishra A, Ochs-Balcom HM, Freundenheim JL, et al. Association between anemia and quality of life in a population sample of individuals with chronic obstructive pulmonary disease. BMC Pulm Med 2006;6:23.
12. Udovičić M, Baždarić K, Petrovečki M, Bilić-Zulle L. What we need to know when calculating the coefficient of correlation Biochem Med 2007;17:10-5.
13. Richmond J, Kiss J. Anemia in COPD: the role of blood transfusion. Transfusion Medicine Update. Available at URL: http//www.itxm.org.tmn 2008/2008issue4.pdf. Accessed February 5, 2009.
14. Pinto-Plata VM, Mullerova H, Toso JF, Feudjo-Tepie M, Soriano JB, Vessey RS. C-reactive protein in patients with COPD, control smokers and non-smokers. Thorax 2006;61:23-8.
15. John M, Lange A, Hoernig S, Witt C, Anker SD. Prevalence of anemia in chronic obstructive pulmonary disease: comparison to other chronic disease. Int J Cardiol 2006;111:365-70.
16. Portillo K, Belda J, Anton P, Casan P. COPD and anemia: an underdiagnosed association. Rev Clin Esp 2007;8:383-7.
17. Shor AF, Doyle J, Stern L, Dolgitser M, Zilberberg MD. Anemia in chronic obstructive pulmonary disease: epidemiology and economic implications. Curr Med Res Opin 2008;24:1123-30.
18. Cote C, Zilbergberg MD, Mody S, Celli B. The prevalence of polycythemia in a chronic obstructive pulmonary disease (COPD) cohort. Chest 2005;128:264S.
19. Sinn DD, Man SF. Why are patients with chronic obstructive pulmonary disease at increased risk of cardiovascular diseases? The potential role of systemic inflammation in chronic obstructive pulmonary disease. Circulation 2003;107:1514-9.
20. Cote C. Haemoglobin level and its clinical impact in a cohort of patients with COPD. Eur Respir J 2007;29:923-9.